Researchers look to the genes behind COVID-19 for early answers.
1:15 PM
Author |
Editor's note: Information on the COVID-19 crisis is constantly changing. For the latest numbers and updates, keep checking the CDC's website. For the most up-to-date information from Michigan Medicine, visit the hospital's Coronavirus (COVID-19) webpage.
Interested in a COVID-19 clinical trial? Health research is critical to ending the COVID-19 pandemic. Our researchers are hard at work to find vaccines and other ways to potentially prevent and treat the disease and need your help. Sign up to be considered for a clinical trial at Michigan Medicine.
Viruses may seem like cunning villains, purposefully mutating to increasingly deadlier forms to outwit their human hosts. In reality, a lot of what happens with a virus is completely random. This randomness can make figuring out where a virus came from, how it spreads and what makes it tick especially tricky. For SARS-CoV-2, the new virus that causes COVID-19, scientists are looking to its genome for answers to some of these questions.
Researchers were recently able to determine that New York City may have been the original epicenter of the U.S. epidemic and that those initial cases were likely imported from Europe. They can tell this by "looking at the genome and its sequence and seeing how they are similar or different," explains Adam Lauring, M.D., Ph.D., associate professor of microbiology and immunology and infectious disease.
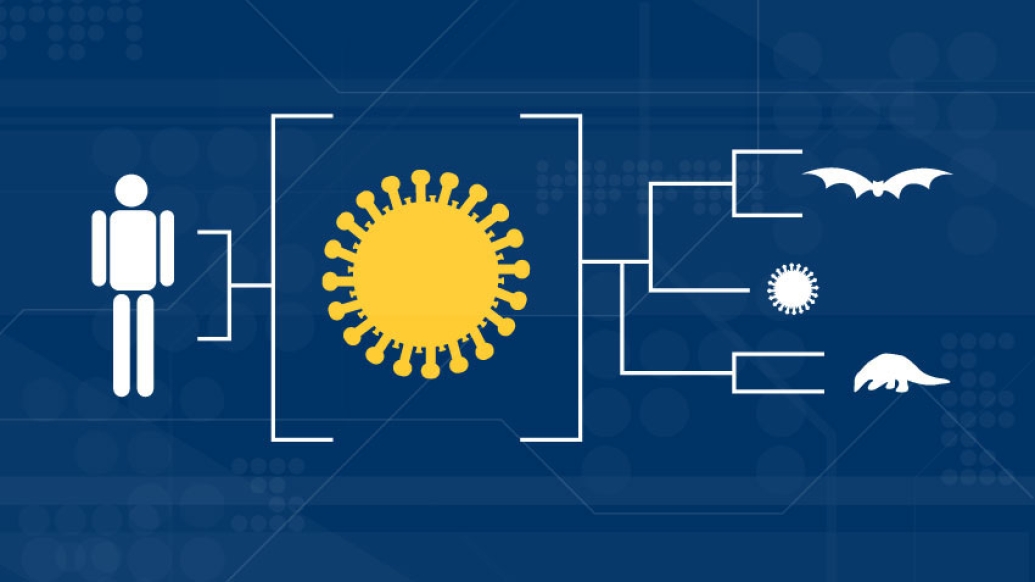
Armed with virus samples taken from people with COVID-19, virologists and epidemiologists create what is known as a phylogenetic tree. This viral family tree lines up the genetic codes from each sample of virus to see who's related to whom.
Based on the genetic sequences and time of collection, you can start to paint a picture of how the virus moves through a population. "The earliest [virus samples] in New York were more similar to the ones from people in Europe who were infected. You start with the dates, then look at the sequences and figure that's the most likely scenario," says Lauring. "It's not foolproof, though. There's always uncertainty."
A real world example of this uncertainty came to light with a study posted online in April, which described the deaths of two people from COVID-19 in Santa Clara, California weeks earlier than the virus was thought to be in California. "What this tells us is that there's definitely missing data," says Lauring. This begs the question, he says, of where did those cases came from and how long the virus was spreading before the outbreak was recognized.
SEE ALSO: Seeking Medical Care During COVID-19
Researchers are also looking at the SARS-CoV-2 genome for clues about its true origin: the animal that infected the first person. So far, bats appear to be the most likely suspect. "Looking at the phylogenetic tree, we see that a bat coronavirus is the closest relative to SARS-CoV-2, sharing around 96% of their genomes," says Lauring. But that too, is not the full story. Another animal, a small, scaly-skinned mammal called a pangolin, has been implicated as well.
"The spike protein in SARS-CoV-2, the main protein on the surface that binds to the cell's receptor and how the virus gets into the cell, is similar to a pangolin coronavirus spike protein," says Lauring. "It's almost like, when you tell a person he has his father's nose. That feature is similar, but across features the father and child may not look very similar. Coronaviruses, like a lot of other viruses, swap genes around."
Attack of the mutants?
These swaps are examples of mutations, which are common in RNA viruses like SARS-CoV-2. Lauring's lab focuses on mutations in influenza, the RNA virus behind the infamous 1918 Spanish flu pandemic. Understanding how influenza mutates is critical for making decisions about the annual influenza vaccine. RNA viruses mutate relatively quickly because they lack a proofreading mechanism to look for and repair errors during replication.
MORE FROM THE LAB: Subscribe to our weekly newsletter
However, SARS-CoV-2 and its coronavirus cousins are unique among RNA viruses, because they have a proofreading enzyme. "The coronavirus genomes are three times longer than you'd expect them to be, and the presence of the proofreading enzyme explains that nicely," says Katherine Spindler, Ph.D., professor in the department of microbiology and immunology. Spindler is a host for the podcast "This Week in Virology," which examines the latest science around SARS-CoV-2 and other viruses.
"With this enzyme, the virus can make a few more errors and not have it be lethal for the virus." As a result, SARS-CoV-2 mutates more slowly than other RNA viruses. Spindler notes that only about 20 mutations have been retained in the genome so far since the beginning of 2020, despite the billions of times the virus has replicated.
SEE ALSO: Keeping Our Patients Safe During COVID-19
Even with its relatively slow mutation rate, mutations present in each person's SARS-CoV-2 genome allows researchers to do genetic tracing in real time, says Lauring. His lab hopes to study the virus' genome more closely to look at how the virus is transmitted in healthcare settings and communities.
He stresses that just because a virus mutates doesn't mean the mutations are making it stronger, more likely to be transmitted, or that it will be tougher to develop a vaccine. "My hunch is evolution won't be the biggest challenge in developing a vaccine. There are viruses that evolve relatively quickly for which we do have vaccines, for example polio, measles, mumps, Ebola, hepatitis A," notes Lauring.
Spindler adds that the fact that we're seeing a variety of COVID-19 symptoms doesn't mean there are different mutant strains. "Every new symptom that comes along, from COVID toes and skin rashes to blood clots, are likely just additional manifestations of the virus as it infects so many different people," she says. Figuring out the mysteries of SARS-CoV-2 will take years of experimental work, she says.
Like Podcasts? Add the Michigan Medicine News Break to your Alexa-enabled device or subscribe for updates on iTunes, Google Play and Stitcher.

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!