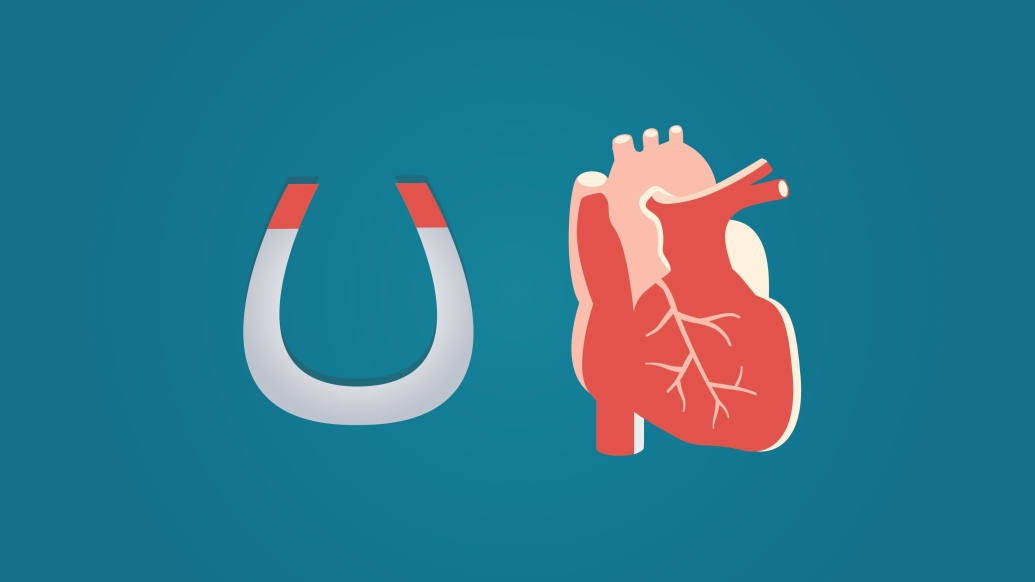
Could magnets be the key to reducing blood clots in LVAD devices? New research compares a different design with the current favorite.
7:00 AM
Author |
Whether used as a bridge to a heart transplant or destination therapy (i.e., permanent therapy), left ventricular assist devices, or LVADs, help end-stage heart failure patients return to an active lifestyle.
MORE FROM THE LAB: Subscribe to our weekly newsletter
The devices are implanted in the body, pumping blood from the left ventricle into the aorta, and connected to a driveline cable that extends outside the body to power them.
"There is a survival benefit and a quality of life improvement," says Francis Pagani, M.D., Ph.D., cardiac surgeon at the University of Michigan's Frankel Cardiovascular Center. "Still, there are important complications with LVADs that need to be addressed."
The largest concern is pump thrombosis, when blood clots in the pump. Thrombosis leads to other complications including stroke and infection. Another problem is gastrointestinal bleeding, in part from the blood thinners LVAD patients receive.
Pagani and Keith Aaronson, M.D., M.S., also of the Frankel CVC, are investigators on the multi-institution MOMENTUM 3 clinical trial that examines whether a new LVAD with a different design would reduce rates of pump thrombosis.
Presented as a late-breaking trial at the American Heart Association's Scientific Sessions 2016, the authors reported preliminary results that the HeartMate 3 was successful in protecting a patient's blood cells from further damage, greatly reducing the risk of blood clots forming in the pump. The results were published simultaneously in the New England Journal of Medicine.
The MOMENTUM 3 trial compares the HeartMate 3 to the current and commonly used HeartMate II LVAD. It examined outcomes including stroke and reoperation rates for the first six months after implantation surgery for 294 subjects (152 with the HeartMate 3, and 142 with the HeartMate II). St. Jude Medical, the manufacturer of both devices, funded the research.

A difference in design
Although the ultimate lifesaving function remains the same, the new design features a significant engineering change: The HeartMate 3 uses a fully magnetically levitated centrifugal-flow pump instead of the HeartMate II's axial continuous-flow pump.
SEE ALSO: PIONEER Study for Afib, PCI Patients Meets Goal
"This design eliminates friction and heat generation from metal contact in the bloodstream," Pagani says. The goal of suspending the rotor in the blood with a magnet is to reduce that stress on the blood and avert pump thrombosis.
Such advancement to the LVAD comes after many years of updates, namely in a significant reduction in pump size — shrinking it from the size of a grapefruit to something more like a golf ball.
That, Pagani says, helps facilitate easier operations with quicker recovery.
"LVADs are also more durable now, and patients have been supported on them for 10 years or more," he adds. "The HeartMate 3 is the next step forward."
Positive preliminary results
None of the subjects who used the new HeartMate 3 pump experienced pump thrombosis during the six-month follow-up period.
By comparison, 14 control-group patients using the HeartMate II were reported to have suspected or confirmed pump thrombosis.
"These results demonstrate that the engineering design works in terms of preventing blood clots from forming in the pump," Pagani says.
The group with the new devices also required fewer reoperations for pump malfunctions, which carry risk for patients.
However, death and disabling stroke rates, along with quality of life reports, were not significantly different between the two groups.
Next steps and advisement
The MOMENTUM 3 trial is another step in determining the future of LVAD design and safety. The HeartMate 3 trial enrolled more than 1,000 subjects, and this initial report addresses just 294 of them over a six-month period.
SEE ALSO: Ticagrelor Not Superior for PAD
"The trial is designed to keep following the subjects for two years," Pagani says. "There is a lot more information that needs to come out of the trial before we make conclusions about the device."
Which is why he maintains that it's safest for existing LVAD patients to keep the one they have even as these studies are presented.
"The operation exchange would carry too high of a risk," Pagani says. "That risk currently outweighs the benefit of a newer pump."
Pagani notes the HeartMate 3 likely won't be available to those not involved in a trial for at least a year or two, pending Food and Drug Administration approval.
Disclosure: Pagani also performs research with HeartWare, the other leading LVAD manufacturer.

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!