From moderate-risk TAVR to PCSK9 inhibitors to exercise in HCM, the American College of Cardiology’s annual Scientific Session brought advancements across many areas of cardiology.
8:00 PM
Author |

One of the most prestigious cardiology conferences, the annual American College of Cardiology's Scientific Session brings experts from around the globe together, presenting and debating the latest research.
MORE FROM THE LAB: Subscribe to our weekly newsletter
Some of the most anticipated results covered exercise benefits for people with hypertrophic cardiomyopathy, extended treatment for venous thromboembolism and a new drug that could drastically lower cholesterol.
Below, Michigan Medicine experts note some of the weekend's standouts.
RESET-HCM
Current guidelines recommend people with HCM, the most common genetic cardiovascular disease, limit intense exercise because of concerns over triggering ventricular arrhythmias. But new Michigan Medicine research, published in JAMA, finds there may be reason to re-evaluate the guidelines.
The preliminary study announced a small but statistically significant increase in exercise capacity in HCM patients who underwent moderate-intensity exercise training.
Researchers studied 136 patients with HCM between ages 18 and 80. For 16 weeks, members of one group were told to continue with their usual level of physical activity while the other group members were given individualized exercise plans the researchers created.
The exercises were moderate, including walking, using an elliptical machine, jogging or biking, and excluding intervals or weight training. The exercise group participants experienced a small but statistically significant increase in peak VO2 max after the 16 weeks. Neither group experienced any major adverse effects.
"The findings show patients that follow an exercise prescription can actually train and improve their functional capacity," says first author Sara Saberi, M.D., of Michigan Medicine's Frankel Cardiovascular Center.
The long-term safety of exercise in the HCM population remains to be established, but the researchers call this study an important first step.
FOURIER, SPIRE 1 and SPIRE 2, EBBINGHAUS
A new class of drugs could lower cholesterol levels by more than half, but are they truly effective in lowering the risk of cardiovascular disease?
Late-breaking clinical trials investigated PCSK9 inhibitor evolocumab, or Repatha, for effectiveness and safety. FOURIER, published in the New England Journal of Medicine, studied 27,564 subjects with atherosclerotic cardiovascular disease.
"FOURIER demonstrated that on top of contemporary medical therapy including statins, evolocumab reduced the primary outcome (combined cardiovascular events) an additional 15 percent over 2.2 years," says Robert Brook, M.D., of U-M, who was not involved in the study. "It is clear that the era of PCSK9 inhibition is here to stay."
But Brook and others note concerns over the high cost of the drugs, and whether the long-term benefits and safety are enough to merit the additional burden.
There have also been questions about memory and cognition among those reaching very low LDL levels with PCSK9 inhibitors. In EBBINGHAUS, 1,204 of the FOURIER patients taking evolocumab underwent cognitive testing. After 20 months, researchers reported no adverse cognitive effects.
SPIRE 1 and SPIRE 2, in the New England Journal of Medicine as well, also addressed PCSK9 inhibitors, though Pfizer discontinued development of bococizumab last November. Researchers reported no benefit for lower-risk subjects, but a significant benefit for higher-risk subjects.
EINSTEIN CHOICE
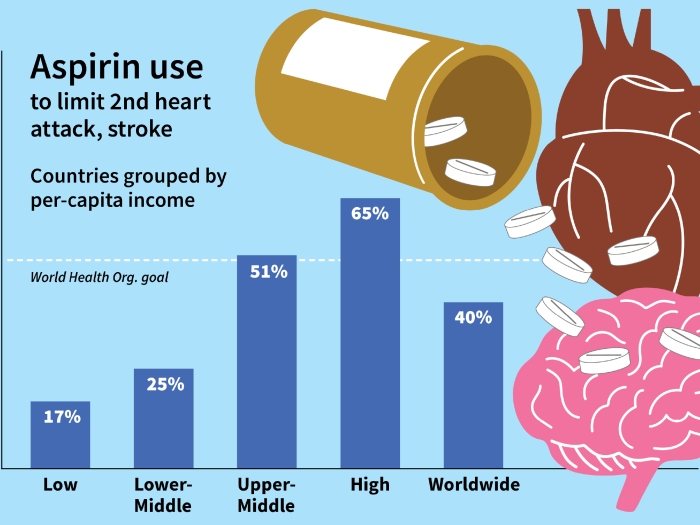
The EINSTEIN CHOICE trial compared aspirin to a newer agent, rivaroxaban, for extended treatment of venous thromboembolism to prevent recurring blood clots in the veins of the legs or in the lungs.
"While blood thinners do a great job preventing recurrence, once you stop a blood thinner the recurrence rate can be very high," says Geoffrey Barnes, M.D., MSc, of U-M, who was not involved in the study. "However, taking blood thinners for many years puts patients at risk of bleeding complications. EINSTEIN CHOICE directly compared two doses of rivaroxaban to aspirin for VTE prevention."
The study, published in the New England Journal of Medicine, determined continuing rivaroxaban, also known as Xarelto, for a year was more successful than aspirin, with less events and no more bleeding.
Barnes notes this trial was similar to AMPLIFY-EXT, published in the New England Journal of Medicine in 2013, which compared apixaban, or Eliquis, at two doses to placebo for extended treatment of VTE.
Jackson Heart Study: Statin guidelines differ for African-Americans
A new study published in JAMA Cardiology determined the stricter statin guidelines did not identify as many African-American individuals with coronary calcification as the current ACC/AHA guidelines.
The 2013 American College of Cardiology and American Heart Association guidelines dramatically increased the number of people eligible for statins. Recent guidelines developed by the U.S. Preventative Services Task Force, or USPSTF, focus on a narrower population using clinical risk factors.
"We found one in four African-Americans who would have been recommended for statin therapy under the ACC/AHA guidelines would not be recommended under new USPSTF guidelines," says Venkatesh Murthy, M.D., Ph.D., co-senior author from U-M.
Murthy led a study of 2,812 African-American adults who participated in the Jackson Heart Study in Jackson, Mississippi, and were at risk for atherosclerotic cardiovascular disease.
"Because the USPSTF guidelines target those with the highest risk of atherosclerotic cardiovascular disease, they also may miss some African-Americans with some vascular calcification and low or moderate risk," says Ravi V. Shah, M.D., co-senior author from Massachusetts General Hospital and Harvard Medical School.
Researchers say the results support further personalization of recommendations for statin therapy, including targeted imaging.
Read more in the Frankel Cardiovascular Center's newsroom.
SURTAVI
One of the most anticipated late-breaking clinical trials, SURTAVI shows a less-invasive valve replacement procedure was not inferior to surgery in patients with intermediate surgical risk.
Transcatheter aortic valve replacement, or TAVR, is typically used for patients with severe aortic stenosis who may have a more difficult recovery from an open heart surgery.
But SURTAVI, published in the New England Journal of Medicine, shows TAVR may also be an aortic valve replacement option for moderate-risk cases. The study, conducted at 87 centers, randomized patients to either undergo either TAVR with a self-expanding bioprosthesis or open surgery.
Two years after the procedures, the rates of death and disabling strokes were similar between groups.
Stanley Chetcuti, M.D., and G. Michael Deeb, M.D., of Michigan Medicine's Frankel Cardiovascular Center, served as investigators on the trial.
Researchers hope to gain FDA approval later this year.

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!