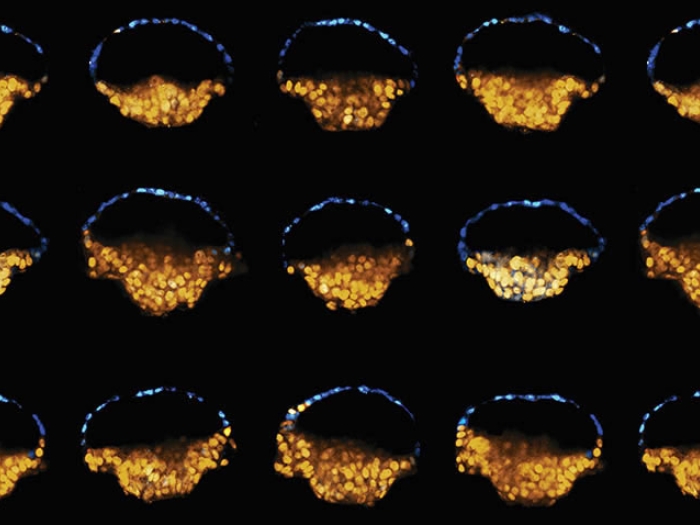
By harnessing massive amounts of data on activity within and between snippets of DNA, researchers aim to one day reprogram both healthy and diseased cells.
1:00 PM
Author |

In fairy tales, all it takes to transform a frog into a prince, a servant into a princess or a mouse into a horse is the wave of a magic wand.
MORE FROM THE LAB: Subscribe to our weekly newsletter
But in the real world, transforming one living thing into another isn't so easy. Only in recent years have scientists discovered how to do so by using tiny individual living cells.
In fact, the team that figured it out won the Nobel Prize for discovering how to transform an ordinary human skin cell into a stem cell — the same kind of cell found in embryos. With painstaking effort, such cells can grow up to become any other kind of cell in the body.
And in the past decade, that time-consuming transformation technique has opened the door for discoveries about many conditions ranging from birth defects to cancer.
Still, what if scientists could cut out a step by eliminating the induced pluripotent stem -cell middle stage to go straight from a skin cell to any other kind of cell?
A new paper in the Proceedings of the National Academy of Sciences lays out a way to do it by harnessing the wealth of data now available about DNA activity and using it to reprogram cells directly.
The formula also provides a blueprint for determining the optimal combination of factors and timing to accomplish this reprogramming. Using this formula, the authors were able to predict the factors the Nobel-winning team discovered, a process that required years of trial and error.
Developed by a team of University of Michigan scientists with colleagues from the University of Maryland and Harvard University, the concept combines biological information on genome structure and gene expression with a fair bit of math — an approach called data-guided control. The paper's authors include world experts on control theory Roger Brockett, Ph.D., of Harvard, and U-M mathematics department chair Anthony Bloch, Ph.D.
Although the authors spell out an algorithm for transforming cells and successfully predict factors that are already known to reprogram cells, they didn't directly test the formula in the laboratory. The team plans to further test the method and hopes that scientists at Michigan and around the world can execute it.
If successful, they predict it could have major applications that include regenerating diseased or lost tissue and fighting cancer.
"Cells in our body naturally specialize," says Indika Rajapakse, Ph.D., a U-M bioinformatics and mathematics researcher and senior author of the new paper. "What we propose could provide a shortcut to doing the same: to help any cell become a targeted cell type."
Cells in our body naturally specialize. What we propose could provide a shortcut to doing the same: to help any cell become a targeted cell type.Indika Rajapakse, Ph.D.
Building on past progress
The idea of direct reprogramming is not new, Rajapakse notes.
In the late 1980s, a team led by the late scientist Harold Weintraub turned skin cells directly into muscle cells by bathing the cells in a type of molecule that encouraged certain genes in the cells' DNA to be "read" and produce molecules that drove changes in the cell. Rajapakse trained with Weintraub's colleague, Mark Groudine, Ph.D., at Fred Hutchinson Cancer Research Center in Seattle.
SEE ALSO: How Stem Cells Grow into Structures That Could Aid Understanding of Fertility
The new model builds on that idea by harnessing the power of these molecules, called transcription factors, or TFs.
But instead of bathing the whole cell culture in one TF, the scientists aim to target cells with specific TFs at crucial times in their life span.
Their updated process relies on a mathematical control model for harnessing all the information that can now be learned about cells at the molecular level and combining it to map out a strategic timing and sequence for injecting TFs to get the desired cell type.
"We have so much data now from RNA and transcription factor activity, and from Hi-C data of chromosome configuration that tells us how often two pieces of chromatin are near one another, that we believe we can go from the cell's initial configuration to the desired configuration," says Rajapakse.
The Hi-C technique lets scientists track the location of — and contact between — portions of the DNA-protein complex called the chromatin. So even if two genes sit far apart on a long strand of DNA, they may come in close contact when those looping, folding strands end up next to each other.
If one of those genes is "read," it may produce a TF that then sets in motion the "reading" of the other gene, and the production of a certain protein that plays a key role in transforming the cell in some way.
The amount of data that would come out of analyzing these chromatin interactions (called topologically associating domains, or TADs) in just one type of cell is huge. But modern bioinformatics techniques make it easier to make sense of it all.
Important implications
The ongoing work represents a team effort across several U-M disciplines.
The first author of the paper is Scott Ronquist, a Ph.D. student who began working with Rajapakse in the Computational Medicine and Bioinformatics Department as a U-M undergraduate. He and former postdoctoral fellow Geoff Patterson, Ph.D., led the effort to use gene expression and TAD data generated in the Rajapakse lab and publicly available gene expression and TF data to test their model. They were able to see patterns in the data that reflected the pace of normal cell differentiation.
Now, they're working on testing the model in the laboratory of Max Wicha, M.D., the Madeline and Sidney Forbes Professor of Oncology at the U-M Comprehensive Cancer Center and the center's former director.
"This algorithm provides a blueprint that has important implications for cancer in that we think cancer stem cells may arise from normal stem cells via similar reprogramming pathways," says Wicha, who is a co-author on the new paper. "This work also has important implications for regenerative medicine and tissue engineering since it provides a blueprint for generating any desired cell type.
"It also demonstrates the beauty of combining mathematics and biology to unravel the mysteries of nature."
NOTE: The university has filed for a patent on the algorithm. The work was funded by the Defense Advanced Research Projects Agency, or DARPA.

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!