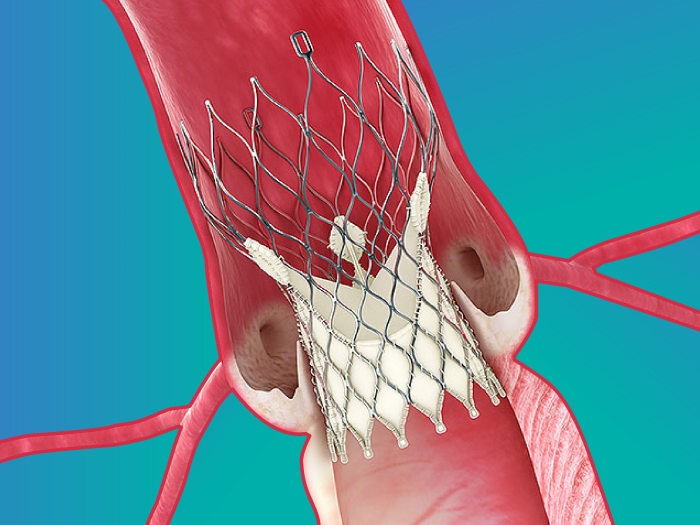
TAVR has changed the landscape for high-risk patients in need of new aortic heart valves. Now, research shows it also has lasting effects.
1:30 PM
Author |
For high-risk patients who need new aortic heart valves — and the doctors who treat them — options used to be in short supply.
The minimally invasive procedure known as TAVR has changed the landscape. Now, new research shows it has lasting effects for high-risk patients.
G. Michael Deeb, M.D., professor of cardiac surgery at the University of Michigan Frankel Cardiovascular Center, presented positive results about TAVR, or transcatheter aortic valve replacement, during a featured clinical research session at the American College of Cardiology Scientific Sessions in Chicago.
In the High Risk Study of CoreValve U.S. Pivotal Trial, Deeb and colleagues compared TAVR using the Medtronic CoreValve System with surgical aortic valve replacement out to three years. TAVR showed superior outcomes. The findings were simultaneously published in the Journal of the American College of Cardiology.
The new data support the viability of TAVR out to three years with no signal of a significant increase in mean gradient or aortic regurgitation.G. Michael Deeb, M.D.
Of the 407 patients studied, 46.7 percent who had TAVR were alive and stroke-free three years later compared with 37.3 percent of patients who had surgery.
Patients treated with the CoreValve System showed superior outcomes compared to surgery for all strokes (12.6 percent vs. 19.0 percent). All-cause mortality continued to be numerically lower for TAVR compared with surgery-treated patients (32.9 percent vs. 39.1 percent).
"It's reassuring to see that the CoreValve High Risk Study continues to show that TAVR is superior to surgery at three years for the combined endpoint of mortality and stroke, which has the most important impact on patients," says Deeb, the Herbert Sloan Collegiate Professor of Cardiac Surgery. "Most importantly, the new data support the viability of TAVR out to three years with no signal of a significant increase in mean gradient or aortic regurgitation."
TAVR allows doctors to reach the aortic valve using hollow tubes called catheters rather than opening a patient's chest during surgery. The procedure is a boon to patients for whom open-heart surgery is too risky.
The U.S Food and Drug Administration approved the CoreValve System in 2014. It was the first TAVR device approved in the United States for valve-in-valve procedures in both high-risk and extreme-risk patients with failed surgical valves.
The surgeons and interventional cardiologists at the U-M Frankel Cardiovascular Center have performed more than 640 TAVR procedures, making the program one of the busiest in the country. U-M researchers participated in clinical trials leading to the TAVR devices on the market today.
"One of the great aha moments in my life was seeing the difference between a post-procedure transcatheter aortic valve patient versus a surgical valve patient," says Deeb. "After the (TAVR) procedure they're ready to go. They want to go home. They want to get back into life. It's that dramatic."

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!