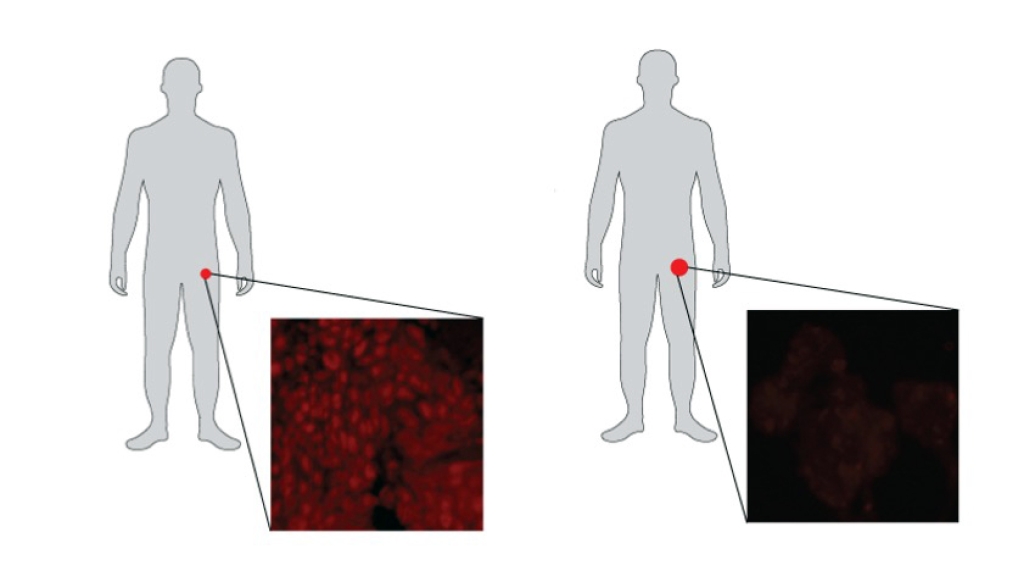
Biopsies from the same patients before and after treatment reveal how a specific drug reprograms prostate tumors.
5:00 AM
Author |
Drugs like enzalutamide that inhibit male hormones from activating the androgen receptor have been used to treat advanced prostate cancer for more than a decade. While successful in most cases, these drugs can eventually stop working, but there is a limited understanding about how this change occurs.
A new study from the University of Michigan Rogel Cancer Center suggests androgen receptor inhibitors can fundamentally rewire and reshape how prostate tumors function, and in certain cases even make them more aggressive. These findings were published in Nature Communications.
Male hormones function as fuel, turning on the androgen receptor that acts as the engine of prostate cancer cells. For the past 80 years, treatment for patients with advanced prostate cancer has focused on interfering with these hormone levels—now typically done through hormone lowering shots and drugs like enzalutamide. Eventually, nearly all tumors develop workarounds and escape treatment, and in most cases, tumors remain dependent on male hormones to power their growth. Other examples of treatment resistance remain poorly understood.
"The greatest unmet need in the clinic right now is understanding the workarounds in a tumor that becomes resistant to androgen receptor targeting drugs so we can determine how best to treat the patient whose tumor has begun to grow," said Joshi Alumkal, M.D., Wicha Family Professor of Oncology and Professor of Internal Medicine, whose team led this research in collaboration with the Zheng Xia laboratory at the Oregon Health & Sciences University Knight Cancer Institute. Thomas Westbrook, M.D., hematology-oncology fellow, was the study's co-first author along with post-doctoral fellow Xiangnan Guan, Ph.D. "Once enzalutamide stops working, there are limited options. We don't know how or why most tumors become resistant."

Alumkal wanted to understand what was present in these tumors to begin with and what happened after tumors started to grow on enzalutamide treatment.
He and colleagues recruited patients to a longitudinal study to obtain metastatic biopsies before enzalutamide treatment and at the time the tumor became resistant to treatment. His team collected serial biopsies from 21 patients, enabling them to understand the workarounds in the tumor from each patient.
Alumkal says this is the largest collection of matched metastatic biopsies before and after enzalutamide.
"To understand resistance to drugs, researchers often collect samples from some patients before treatment and from a different group of patients whose tumors are treatment resistant. However, that approach is much less precise because there could be other significant differences between those patients. You can't pinpoint if the differences have anything to do with drug exposure or have more to do with the tumors just being different to begin with."
Alumkal's sequential sampling method provided a much clearer picture of how enzalutamide resistance might emerge.
When they compared the baseline sample to the progression sample from the same patient, most tumors showed no significant gene expression changes.
"That the gene expression program of a tumor prior to treatment looked very similar at progression while on enzalutamide is quite remarkable," Alumkal said. "It speaks to how well most of the tumors were able to adapt and keep the androgen receptor engine on despite enzalutamide treatment."
But that wasn't the only surprise.
In three of the 21 cases, Alumkal and his team saw a profound shift in the wiring—or gene expression program—of the tumors.
"We knew that sometimes tumors become fuel-independent and no longer rely on the androgen receptor. These tumors instead turn on a gene expression program more common in nerve cells, rather than prostate cells, and shift to an aggressive form called neuroendocrine prostate cancer."
But Alumkal found that in 15% of cases, the tumors also became fuel-independent for another reason.
"These tumors were wired in a unique way and were most consistent with a subtype of prostate cancer called double-negative prostate cancer, meaning the tumors no longer had the androgen receptor as an engine. But they also did not become neuroendocrine prostate cancer."
Alumkal uses vehicles to describe this change.
"Initially, nearly all prostate tumors are gas guzzlers: very fuel dependent and powered by the androgen receptor as the engine. When treated with hormonal treatments, most tumors remain fuel-dependent but become more fuel efficient, able to go farther with less gasoline.
"Our work showed that the majority of the tumors—even after receiving enzalutamide—remain very fuel-dependent, which suggests that continuing to target the androgen receptor could make an enormous difference in these tumors," Alumkal continued.

Alumkal found that three tumors converted to become double negative prostate cancer—akin to an electric vehicle.
"The gasoline engine was replaced by a completely distinct set of machinery that allowed tumors to grow and survive," Alumkal explained.
The DNA mutations found in the baseline and progression biopsies from these converter tumors were the same, which strongly suggests that enzalutamide completely rewired the engine of the original fuel-dependent tumor to become fuel-independent at disease progression.
"It's a dramatic shift to wrap your head around."
Although the baseline tumors appeared similar under the microscope, Alumkal's team identified specific genes that were highly expressed in those that eventually became double negative prostate cancer. This result suggests that certain tumors exist in a hybrid state, initially dependent on fuel but at risk for becoming a fuel-independent double negative prostate cancer during enzalutamide treatment.
Alumkal says results from the sequential sampling method suggest that enzalutamide is causing tumors to adapt, in some cases dramatically.
Alumkal notes that the gene signature he identified is preliminary, and the team has more work to do.
"Still, the fact that the DNA looks similar in the converters strongly indicates that enzalutamide is reprogramming tumors. We have more work to do, but it may be possible up-front to identify patients at greatest risk of having their tumor become fuel-independent after treatment with drugs like enzalutamide," he said.
Funding: This research was supported by the Stand Up to Cancer-Prostate Cancer Foundation (PCF) Prostate Dream Team Translational Cancer Research Grant SU2C-AACR-DT0409 and a Prostate Cancer Foundation Challenge Award. Other support includes: National Cancer Institute (NCI) R01 CA251245, R01 CA234715; The Pacific Northwest Prostate Cancer Specialized Programs of Research Excellence (SPORE) NCI P50 CA097186; the Michigan Prostate SPORE NCI P50 CA186786; NCI P01 CA163227; the Drug Resistance and Sensitivity Network NCI U54 CA224079 and NCI P50 CA186786-07S1; NCI T32 CA009357; University of Michigan Rogel Cancer Center Innovation Award NCI P30 CA046592; Department of Defense Idea Award (W81XWH-20-1-0405); Department of Defense Idea Award (W81XWH2110539); Grant 2021184 from the Doris Duke Charitable Foundation; the V Foundation; National Comprehensive Cancer Network (NCCN)/ Astellas Pharma Global Development Award; the Sheppard Family Fund.
Conflict of Interest: O. Witte currently has consulting, equity, and/or board relationships with Trethera Corporation, Kronos Biosciences, Sofie Biosciences, Breakthrough Properties, Vida Ventures, Nammi Therapeutics, Two River, Iconovir, Appia BioSciences, Neogene Therapeutics, 76Bio, and Allogene Therapeutics outside of submitted work. T.M. Beer reports consulting fees from AbbVie, Arvinas, Astellas Pharma, AstraZeneca, Bayer, Constellation, Grail Inc., Janssen, Myovant Sciences, Pfizer, Sanofi, Sapience Therapeutics, Bristol-Myers Squib, Novartis, Clovis Oncology, stock ownership in Arvinas Inc, Salarius Pharmaceuticals and researching funding from Alliance Foundation Trials, Astellas Pharma, Bayer, Boehringer Ingelheim, Corcept Therapeutics, Endocyte Inc./Advanced Accelerator Applications (AAA), Freenome, Grail Inc., Harpoon Therapeutics, Janssen Research & Development, Medivation Inc., Sotio, Theraclone Sciences/OncoResponse, Zenith Epigenetics, all outside of submitted work. J. Alumkal has received consulting and speaker's fees from Astellas Pharma, consulting fees from Dendreon, consulting fees from Merck, consulting fees from Bristol Myers Squibb, and research support to his institution from Astellas Pharma, Zenith Epigenetics, and Beactica. The remaining authors declare no conflicts of interest.
DOI: 10.1038/s41467-022-32701-6
MORE FROM THE LAB: Subscribe to our weekly newsletter
Like Podcasts? Add the Michigan Medicine News Break on iTunes, Google Podcasts or anywhere you listen to podcasts.

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!