U-M researchers discover that the very cells that make up lymphoid organs may help HIV spread to T cells more efficiently.
7:00 AM
Author |
Just as walking through the wooden frame of a house allows you to more easily picture the final product than just looking at a floor plan, three-dimensional models can help researchers mimic biological processes better than flat ones in a petri dish.
LISTEN UP: Add the new Michigan Medicine News Break to your Alexa-enabled device, or subscribe to our daily audio updates on iTunes, Google Play and Stitcher.
Akira Ono, Ph.D., associate professor in the University of Michigan Department of Microbiology and Immunology, had this fact in mind when he approached researchers in U-M's Department of Biomedical Engineering to help him model a discovery about how HIV spreads inside the human body.
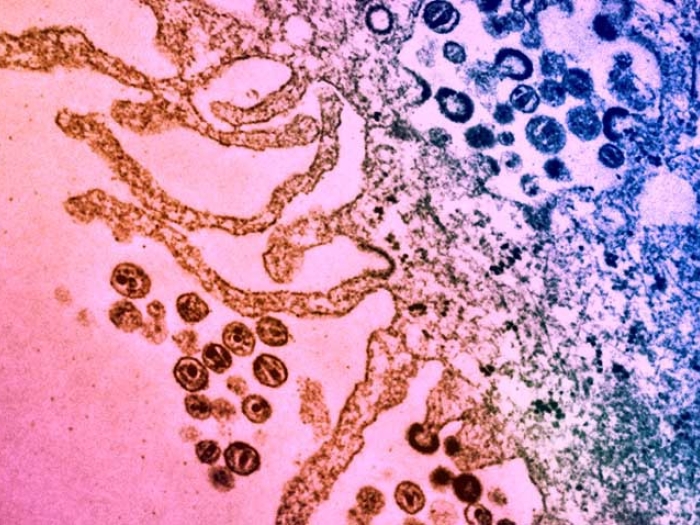
To make the discovery, published in Nature Communications, Ono and his co-author, postdoctoral fellow Tomoyuki Murakami, Ph.D., asked how HIV-1 spreads from T cell to T cell inside lymphoid organs. Lymphoid organs, such as lymph nodes and tonsils, are where more than 90 percent of the T cells in our body are located. T cells are important in regulating the immune system and fighting off infection and are the cells infected and killed by HIV.
But lymphoid organs are made of more than just T cells. T cells are accompanied by cells called fibroblastic reticular cells (FRCs) that form the structure of T cell-enriched zones in the lymphoid organ. His team wondered if this scaffolding might actually be playing a role in the spread of HIV-1.
"When you grow T cells in a dish and introduce HIV-1, the virus will float around and eventually infect the cells when they come into contact," Ono says. "However, when we mixed FRCs in the tissue culture, we saw a much faster spread of HIV." Somehow, the FRCs were capturing the virus without becoming infected themselves. Their team found, for the first time, that the FRCs were then transmitting the virus to the T cells in a process called trans-infection.
When we mixed FRCs in the tissue culture, we saw a much faster spread of HIV.Akira Ono, Ph.D., Department of Microbiology and Immunology
2-D to 3-D
It was one thing to look at cells floating in or sticking to the bottom of a dish. But hard plastic is not necessarily the best model for living tissue.
MORE FROM THE LAB: Subscribe to our weekly newsletter
Ariella Shikanov, Ph.D., associate professor of biomedical engineering, has been working on 3-D models to help investigators like Ono test their observations in a more realistic way. "In the past 30 years, scientists have begun to recognize the value in developing 3-D cultures, which allow you to have better control over your tissue culture environment and mimic physiological properties," she says.
For this experiment, Shikanov developed a matrix made out of an enriched gel and seeded FRCs into it. While in a flat dish-based tissue culture, the cells had a misshapen, spread-out appearance, in the 3-D gel, they grew into a distinctive dense network similar to what is seen in lymphoid organs.
Furthermore, T cells, which also behave differently in 2-D versus 3-D environments, were able to bind to the web-like network formed by the FRCs within the gel block.
Once this network was formed, the Ono and Murakami's team introduced HIV and infected T cells to confirm that, in three dimensions, FRCs are mediating trans-infection of T cells. Shikanov notes, "There is no cure for HIV and developing animal models is expensive and difficult. Being able to develop these models cheaply is very important for progressing our understanding of the disease."
Her lab's next goal is to take these observations to another level. "We are working on using the 3-D model to culture and incubate T cells for longer periods of time to see different stages of infection and maybe see how the virus mutates," says Shikanov.
Capturing the virus
Beyond demonstrating that FRCs were helping infect T cells with HIV, Ono's lab also found two molecules that make it possible — molecules that might be possible targets for drugs against the virus. "These two molecules, CD44 and hyaluronic acid, are binding to each other and thereby bridging the virus particles and the FRCs." His team looked at human tonsil cells and saw that HIV with more CD44 protein spread to T cells from FRCs better than HIV without the protein.
Ono explains, "When we used an antibody to block CD44 or cut hyaluronic acid with an enzyme in tissue cultures, we could reduce this trans-infection activity. If we could do something similar with infected individuals, such treatment may suppress HIV spread in lymphoid organs. Obviously, however, before getting to that stage, we will need to perform more analyses in various models including the 3-D model and animals."

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!