Michigan Medicine researchers investigate how malfunctioning proteins lead to ALS and dementia.
1:00 PM
Author |
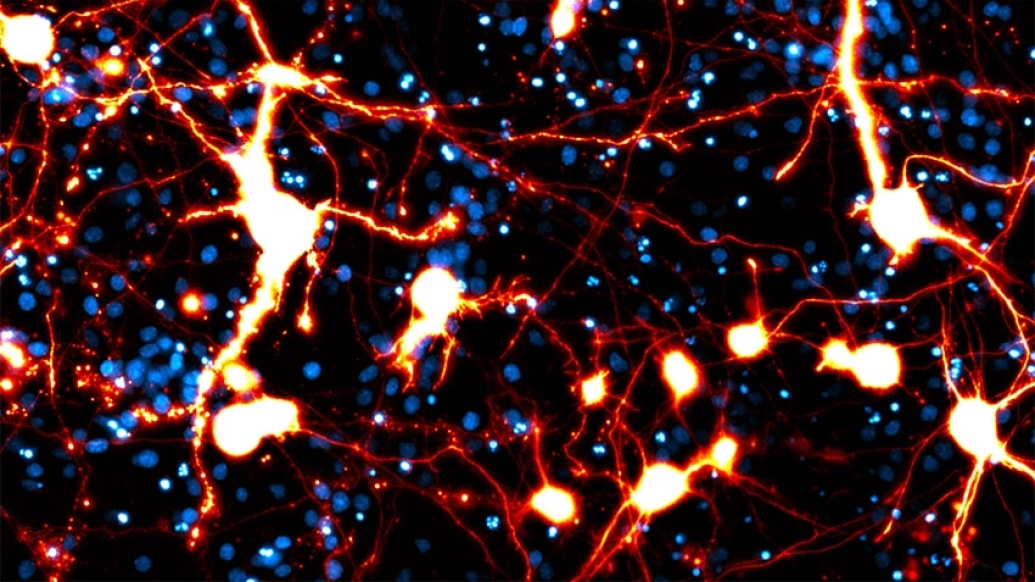
Amyotrophic lateral sclerosis, or ALS, afflicts the body's nerve cells. The rare but devastating disease leads to progressive muscle weakness that can eventually disrupt a person's ability to walk, talk and even breathe.
LISTEN UP: Add the new Michigan Medicine News Break to your Alexa-enabled device, or subscribe to our daily audio updates on iTunes, Google Play and Stitcher.
About a third of people with ALS also have profound changes in their personality due to the destruction of neurons in the brain, developing what is known as frontotemporal dementia, or FTD.
In 2006, scientists discovered a crucial link: Most patients with ALS and FTD had deposits of a protein called TDP-43 in the wrong location in their cells.
Research investigators Elizabeth Tank, Ph.D., Claudia Figueroa-Romero, Ph.D., Sami Barmada, M.D., Ph.D., of the Department of Neurology at the University of Michigan Medical School, and their team have since set out to pinpoint what this protein was doing to prompt these severe conditions.
Their findings, published in the journal Nature Communications, point to a mishap in the regulation of RNA.
RNA's main function is to translate the instructions within a cell's DNA to build proteins. RNAs have varying life spans during which they are translated into proteins, and last from a few minutes to a few days in humans. Once that period is up, the RNA is degraded. The ability of an RNA molecule to resist degradation is called stability.
As an RNA-binding protein, TDP-43 helps regulate the stability of RNA. But how it does so remains unknown.
"We know that TDP-43 is absolutely essential for RNA processing," says Barmada. "Too much TDP-43, and cells die. Too little TDP-43, and they die. If TDP-43 is in the wrong place, they die.
"It has to be in the right place, at the right time, and in the right amounts."
Finding patterns in data
To test whether an overabundance of TDP-43 was affecting RNA stability in patients with ALS and FTD, the team used a technique developed by Mats Ljungman, Ph.D., associate professor of radiation oncology and environmental health sciences in U-M's School of Public Health, to look at the stability of more than 20,000 RNA transcripts in skin cells donated by patients with ALS.
MORE FROM THE LAB: Subscribe to our weekly newsletter
The data revealed a consistent pattern. Most of the RNA that was destabilized in ALS cells seemed to fall into two categories: pathways for energy production in mitochondria and protein creation through ribosomes.
"These are really critical, basic pathways needed for cellular survival," notes Barmada.
To confirm that this pattern wasn't just a fluke found in skin cells, the team turned the skin cells into stem cells, which can become any cell in the body. With the help of colleague and neurology professor Eva Feldman, M.D., Ph.D., they were also able to look in the brains of patients with ALS and FTD, supplied by the U-M Brain Bank and the U-M ALS Repository.
The same pattern involving energy and protein production was found in each cell type they examined.
Trouble increases with age
Still, researchers wondered, if the protein regulation machinery is defective in other types of cells, why does it lead to the destruction of neurons in particular?
Because adult neurons do not divide and are longer lived, they may be more vulnerable to abnormal energy and ribosome production, Barmada hypothesizes. This may also help explain why ALS and FTD are most often diagnosed after age 50.
Barmada believes that the body's cells are able to compensate for malfunctioning protein synthesis by ramping up their protein production. In fact, in ALS cells they saw more ribosomes despite the instability of the RNA.
"It could mean there are problems in protein translation and energy production that individual cells are coping with their entire lifetimes," Barmada says. "Maybe the cells are able to compensate for decades, until one day those compensatory mechanisms fail and disease begins."
'Goldilocks' proteins offer clues
Besides TDP-43, mutations in several other RNA-binding proteins are found in people with ALS and FTD.
One such protein, called Matrin3 (MATR3), is also involved in a muscle disease called distal myopathy. As with TDP-43, MATR3 participates in RNA processing and stability, but is also able to bind DNA.
SEE ALSO: New CRISPR-Cas9 Tool Edits Both RNA and DNA Precisely
Ahmed Malik, a graduate student working with Barmada, investigated the connection between MATR3 mutations and the degeneration of neurons. Their findings are published in a recent issue of eLife.
Malik inserted MATR3 into brain cells from rodents to see what would happen. He observed that too much MATR3, particularly within the nucleus of neurons, is toxic for cells. The effect was even worse for the mutated version of the protein.
Similarly, removing MATR3 was also bad news for the cells. (Much like in the tale of Goldilocks, the balance of this protein needs to be just right.)
He then found that removing MATR3's ability to bind to RNA leads to the formation of liquid-like droplets within the nucleus of neurons, much like oil in water. MATR3 mutations change the viscosity of these droplets, turning it from a honey-like consistency to that of lard.
However, unlike with TDP-43, clusters of MATR3 in the cytoplasm did not have a negative effect.
Instead, MATR3 wreaks havoc inside the nucleus of the cell. Removing the ability of MATR3 to bind DNA also eliminated its toxicity, suggesting that MATR3 somehow interferes with DNA or prevents the transcription of DNA to RNA.
Their team is now trying to figure out precisely what MATR3 is binding to inside the nucleus and how that leads to neuron loss and eventually symptoms of ALS and FTD

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!