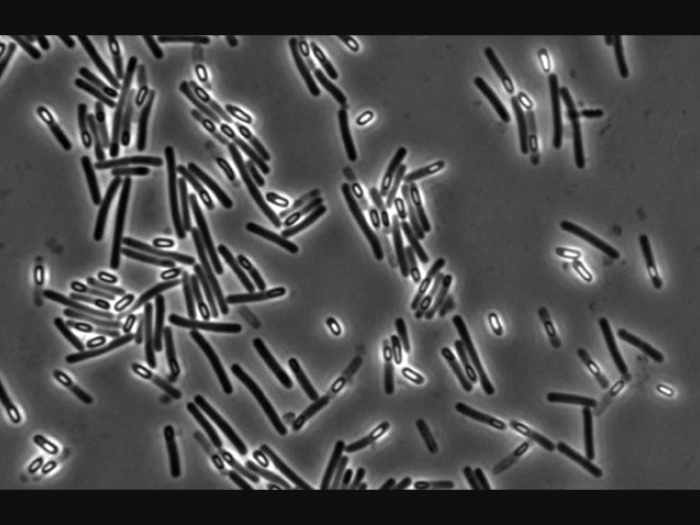
How E coli. and other illness-causing bugs get past the microbiome’s defenses.
5:00 AM
Author |
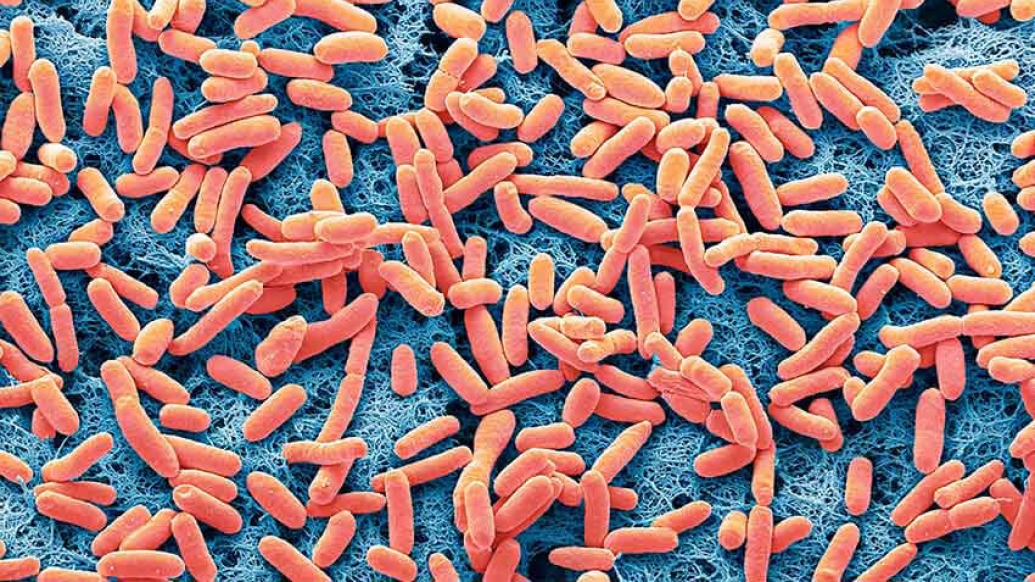
Diarrhea can be the unfortunate but short-lived side effect of bad takeout food, yet it is also a leading cause of death in children under five globally. Scientists continue to investigate how illness-causing bacteria gain a foothold within the digestive tract when they are usually held in check by the protective bacteria collectively known as the gut microbiome.
"Since the 50s, we've known that these beneficial bacteria provide protective responses, one being protection from colonization by pathogens entering via the oral route," says Gabriel Nunez, M.D., the Paul de Kruif Endowed Professor of Pathology at the University of Michigan Medical School.
In a new paper in the journal Cell Host & Microbe, he and lead author Gustavo Caballero-Flores, Ph.D., and their colleagues used a mouse model to look more closely at how bad bacteria overcome the gut's protective mechanisms against pathogen colonization.
LISTEN UP: Add the new Michigan Medicine News Break to your Alexa-enabled device, or subscribe to our daily updates on iTunes, Google Play and Stitcher.
While several previous studies have examined infection in the presence of inflammation (in the case of IBS, for example), the team used a bacteria called Citrobacter rodentium, a mouse pathogen related to the human pathogen Escherichia coli, to analyze how the bacteria mount their initial assault in a non-inflamed, normal gut. E. coli is infamous for several recent food-borne illness outbreaks in romaine lettuce and other foods.
The team created close to 50,000 mutations within the genes of C. rodentium to see which pathogen genes were necessary to overcome the microbiome and colonize the mouse gut. They then exposed uninfected mice and mice bred to completely lack a protective microbiome, called germ-free mice, to compare bacterial colonization with the mutant strains. Using a high-throughput screening method, they found that the genes that were required for colonization in the presence of the microbiome, but not in the germ-free mice, were the ones used to make amino acids, which are the building blocks of proteins.
"Our microbiota give us protection from infection, but themselves benefit by eating our food, including protein from sources like meat and nuts," and by doing so the beneficial bacteria limit the availability of key nutrients such as amino acids to harmful pathogens entering the gut, says Nunez. "A few pathogenic bacteria overcome competition with our microbiome for amino acids in the gut by making their own."
To further test this theory, they fed the mice a high protein diet and found that it helped the pathogenic bacteria to take over more readily. Nunez notes that under normal conditions, a little bit of pathogenic E. coli is not likely to make someone sick. "It requires a high dose of bacteria—the type you get from leaving mayonnaise sitting out in the hot sun—to overcome the microbiome." However, Nunez' team showed in an earlier study that infants and young children lacked this protection, due to their immature microbiomes. "That's one of the reasons we think children are very susceptible to this infection."
While today most bacterial infections are treated with antibiotics –which wipe out good and bad bacteria alike—Nunez notes that the mechanism they describe is likely essential for the survival of the species.
MORE FROM MICHIGAN: Sign up for our weekly newsletter
"Antibiotics were discovered less than 100 years ago. The fact that it takes 1,000,000 bacteria to gain an infection is important. In germ-free mice, 10 bacteria are enough to kill."
Paper cited: "An enteric pathogen subverts colonization resistance by evading competition for amino acids in the gut", Cell Host & Microbe. DOI: 10.1016/j.chom.2020.06.018

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!