A team of researchers conduct a meta-analysis of studies to assess the safety of a commonly used contrast agent for patients with chronic kidney disease.
5:00 PM
Author |
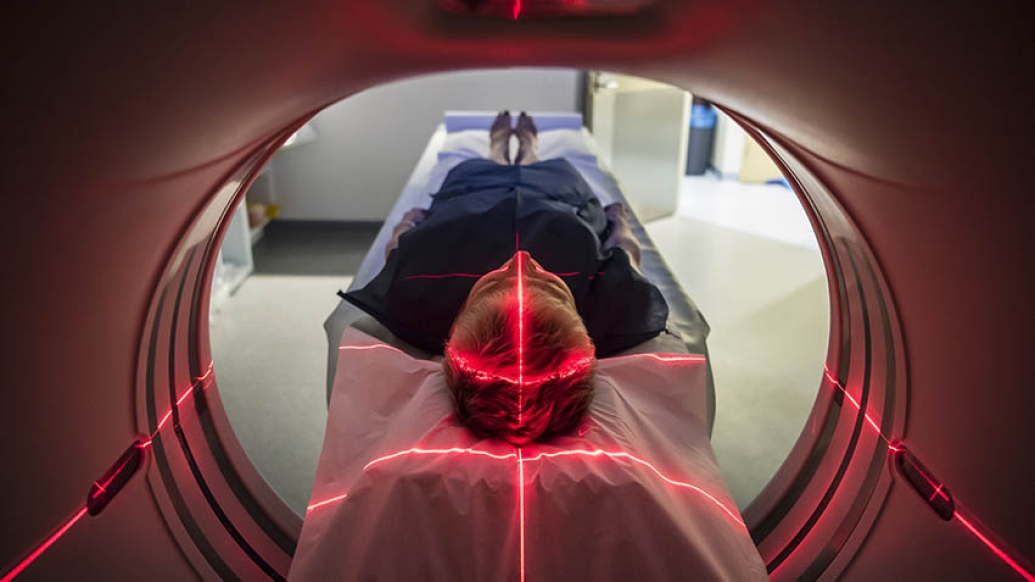
When patients undergo magnetic resonance imaging, they're often given a contrast agent, a substance which helps increase the contrast of bodily structures and fluids, making them easier to see.
According to the United States Food and Drug Administration, gadolinium containing contrast agents (GBCAs) can be defined as intravenous gadolinium-containing drugs used in diagnostic imaging procedures to enhance the quality of MRI or magnetic resonance angiography.
Historically, individuals with chronic kidney disease would often be exposed to these agents during treatment because they served as an alternative to traditional iodinated contrast, which has been linked to the development of nephropathy.
MORE FROM THE LAB: Subscribe to our weekly newsletter
It was eventually reported that upon exposure to GBCAs, some patients developed nephrogenic systemic fibrosis, a debilitating and potentially lethal disease that hardens and thickens the skin and internal organs. In response, the FDA took action and provided recommendations on the population receiving GBCAs.
"However, as time went on, our knowledge about these agents evolved, and we realized the risk is not uniform across all agents," says Matthew Davenport, M.D., an associate professor of radiology and urology and chair of the American College of Radiology Committee on Drugs and Contrast Media.
Our work is not the end of the story. Although we know the risk of developing NSF in patients with CKD is low, we don't know how low it really is. Larger studies will answer that question.Matthew Davenport, M.D.
The American College of Radiology has formally recognized three distinct classes of GBCAs, which are referred to as group II GBCAs. These agents pose the lowest risk for developing nephrogenic systemic fibrosis and may now be used without pre-screening for kidney disease in patients that need a contrast enhanced MRI.
"However, many physicians still steer clear of using group II GBCAs when treating individuals with CKD," says Prasad Shankar, M.D., an associate professor of radiology. "The stigma is there, especially for patients receiving dialysis."
This notion inspired Shankar, Davenport and a collaborative team of clinical and research experts at Michigan Medicine to conduct a meta-analysis to evaluate the risk for developing nephrogenic systemic fibrosis in patients with stage 4 and 5 CKD upon receiving group II GBCAs. Their study also included patients undergoing dialysis.
The results were recently published in JAMA Internal Medicine.
The meta-analysis
The goal of the research team's study was to assess the risk of nephrogenic systemic fibrosis in high risk patients (with severe kidney disease) receiving a low risk group II GBCA.
The study team evaluated 2,700 research papers to identify a final cohort of 16 studies and 4,931 patients. Each of these patients met the defined inclusion criteria for the study's parameters.
LISTEN UP: Add the Michigan Medicine News Break to your Alexa-enabled device, or subscribe to our daily updates on iTunes, Google Play and Stitcher.
The team discovered that across this population, the risk of developing nephrogenic systemic fibrosis from a group II GBCA in patients with stage 4 or 5 CKD was 0%, with an upper bound confidence limit of 0.07% (one side of a two sided 95% confidence interval).
"We found that the per-patient risk of nephrogenic systemic fibrosis from group II GBCA administration in these high risk patients was likely less than 0.07%," says Shankar. "And his study will allow physicians the opportunity to feel more confident when diagnosing disease with MRI in patients with CKD."
"Our work is not the end of the story. Although we know the risk of developing nephrogenic systemic fibrosis in patients with CKD is low, we don't know how low it really is. Larger studies will answer that question," adds Davenport. "We're looking forward to the future."

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!