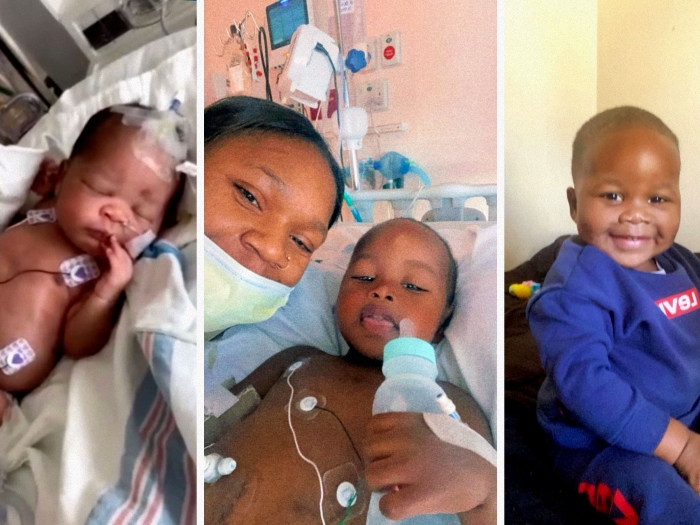
A high-risk gene variant for kidney disease may predict the severity of a child’s nephrotic syndrome, and eventually lead to targeted treatments and screening protocols.
8:00 AM
Author |

When it comes to determining the cause of a black child's glomerular kidney disease, and what the future may hold for that child's health, the presence of a high-risk gene variant could hold a critical clue.
A high-risk variant in the gene known as apolipoprotein L1 (APOL1) has been associated with an increased risk of chronic kidney diseases. The APOL1 variant's strongest association is with focal segmental glomerulosclerosis, a form of nephrotic syndrome, which affects the kidney's filtering ability and causes bodily swelling due to excess protein in the urine.
Past studies of this APOL1 variant, which was first identified in 2010, have focused on adult patients.
But a new analysis from University of Michigan's C.S. Mott Children's Hospital sought to examine how younger people with nephrotic syndrome who also possess the variant might be affected.
The paper considered the collective histories of 104 APOL1 genotyped black children participating in two ongoing studies: the Chronic Kidney Disease in Children (CKiD) cohort and the Nephrotic Syndrome Study Network (NEPTUNE). Results were published in the journal Nephrology Dialysis Transplantation.
The basic assumption that the majority of black children with nephrotic syndrome carried the APOL1 high-risk variant was correct. Although high-risk youths in the studies were diagnosed with nephrotic syndrome later in life compared to affected children without the APOL1 variant, their symptoms were often more severe and advanced more quickly.
"The totality of our research is telling us that these kids seem to be sicker at baseline," says Matthew Sampson, M.D., an assistant professor of pediatrics and a pediatric nephrologist at C.S. Mott Children's Hospital.
"They have a steeper decline of kidney function over time that gets worse more quickly than those who have a low-risk genotype."
The cited studies weren't designed together and did not share the same standardized protocol.
But Sampson, who was the senior author of this analysis and runs the namesake kidney-focused Sampson Lab at U-M, says the data provides an important starting point.
"If we can identify the molecular mechanisms," Sampson says, "we can target therapeutic treatments to help."
If we can identify the molecular mechanisms, we can develop targeted therapeutic treatments to help.Matthew Sampson, M.D.
Looking for clues
The high-risk variant of APOL1, Sampson says, developed among blacks in Africa tens of thousands of years ago as a means to protect against certain types of parasitic illness.
Such genetic defense mechanisms, researchers later discovered, harbor a downside: an increased risk of kidney disease. The variant is not found in white individuals.
One common thread stood out in the CKiD and NEPTUNE data: high-risk APOL1 patients in both studies were far more likely to have been born prematurely.
Other factors, though, were less indicative.
Youths classified as low-risk (those not possessing APOL1) in the studies showed little difference in gender balance, height or body mass index compared to their high-risk counterparts. Nor did socioeconomic factors, such as a parent's age or level of education, range widely between either group.
"We have to identify ways this genomic understanding is useful for our patients," Sampson says.
The next step
Treatment for children with nephrotic syndrome typically involves steroids, Sampson says.
However, in a number of children, steroids are not an effective treatment and only expose these children to harmful side effects. In addition, previous research from the Sampson lab suggested that steroids are not effective in nephrotic syndrome patients with the APOL1 risk genotype. Yet successful treatment is crucial to a patient's health.
A progression of nephrotic syndrome can spur renal failure that will ultimately require dialysis or a kidney transplant.
Might the results of Sampson's analysis lay the groundwork to push for genetic testing at an early age?
Not necessarily, Sampson says.
"Right now, it is not being used clinically at this point in time, either for screening or diagnosis," he says, noting that the likelihood of danger for healthy people in the community with the APOL1 variant remains relatively low. "There are so many people who exist with the high-risk genotype who don't develop kidney disease."
However, the result of the U-M analysis suggests that future studies about the risk of kidney disease may want to focus on black babies with the APOL1 risk variant who were born prematurely.
If it is in fact found that there is an increased risk of kidney disease in this particular group of children, those individuals could receive targeted primary care to detect potential signs of future trouble.
"If such an association is eventually discovered, more frequent monitoring of these children and adherence to standard therapies may be warranted," Sampson says.

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!