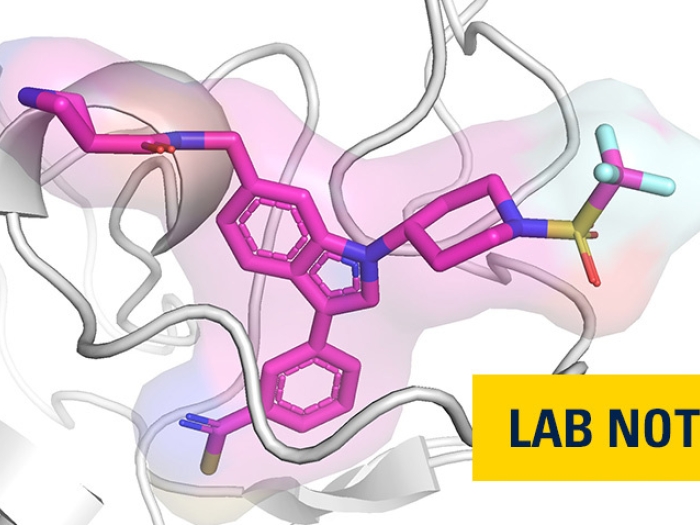
Activity of the polycomb repressive complex 1 is essential for the development and maintenance of leukemic cells; disrupting it presents a new potential therapeutic approach.
11:34 AM
Author |
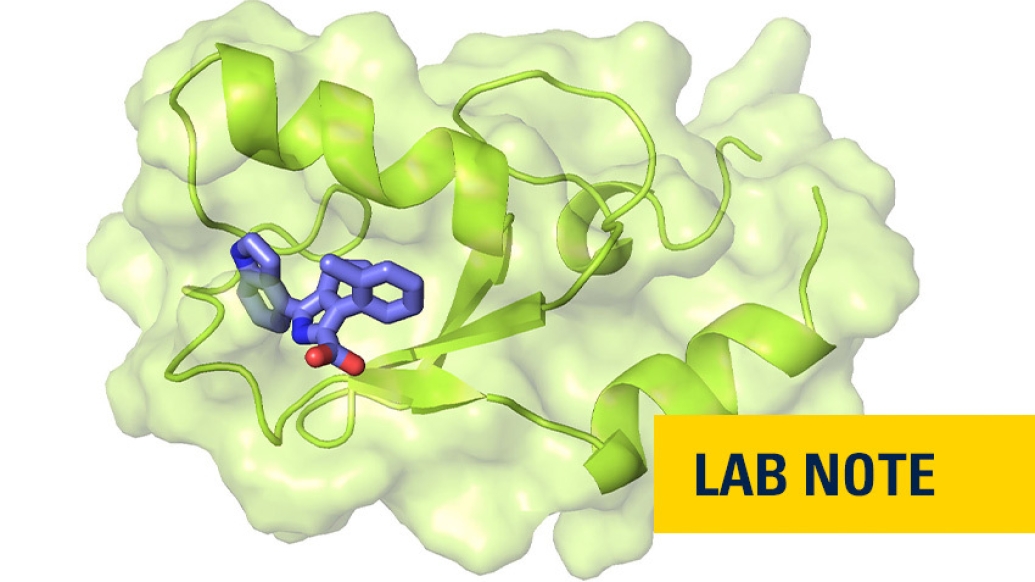
Leukemia stem cells are rare cells that can renew themselves while continuing to generate malignant cells known as leukemic blasts. These cells are difficult to eradicate using chemotherapy drugs and frequently lead to recurrence of leukemia.
Leukemia stem cells, however, are dependent on a protein complex called polycomb repressive complex 1, or PRC1, which interacts with chromatin and turns genes off.
Now a team of researchers led by Tomasz Cierpicki, Ph.D., and Jolanta Grembecka, Ph.D., from the University of Michigan has developed the first small-molecule inhibitors of PRC1 — a first step toward developing a potential new therapeutic approach to treating acute myeloid leukemia by shutting down the activity of leukemia stem cells.
These inhibitors demonstrate activity in leukemia cells and patient samples, as reported in Nature Chemical Biology, and also open new opportunities to study the development of leukemia at the molecular level.
"Our lead compound, RB-3, represents an attractive and unique agent for studying PRC1 biology," said Cierpicki, an associate professor of biophysics and pathology at U-M. "This work demonstrates that directly targeting the activity of PRC1 is indeed feasible and could lay the groundwork for the development of new pharmaceutical agents for leukemia and possibly other cancers."
The study was a close collaboration between his lab and the lab of co-senior author Grembecka, an associate professor of pathology at Michigan Medicine and co-director of the developmental therapeutics program at the U-M Rogel Cancer Center.
Disclosure: The University of Michigan has applied for patent protection on the PRC1 inhibitors and several of the authors are listed as co-inventors.
Paper cited: "Small molecule inhibitors targeting Polycomb repressive complex 1 RING domain," Nature Chemical Biology. DOI: 10.1038/s41589-021-00815-5

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!