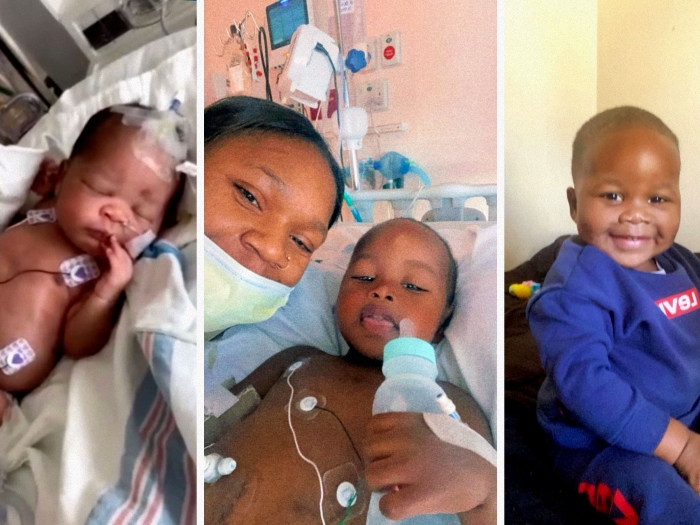
Trial focuses on whether drug udenafil can help prevent chronic liver problems in kids who require a Fontan procedure for congenital heart disease.
3:30 PM
Author |

Some children with complex congenital heart disease require an open-heart surgery known as the Fontan to reroute their body's blood flow, but undergoing the procedure means eventually experiencing some level of liver disease.
While reasons for Fontan-associated liver disease are unclear, experts believe the altered circulation from the procedure may impact blood flow throughout the liver and contribute to liver scarring.
Now, researchers from the Pediatric Heart Network are studying a potential solution to the prevalent complication.
Michigan Medicine teams presented early results from a trial testing whether drug udenafil can reduce liver stiffness that contributes to Fontan-related liver disease in children at the American Heart Association scientific sessions in Chicago.
"There are no treatments for Fontan-associated liver disease and it's universally present in this group of patients," said lead researcher Kurt Schumacher, M.D., a pediatric cardiologist at University of Michigan Health C.S. Mott Children's Hospital, of Michigan Medicine.
"Udenafil relaxes the pulmonary arteries allowing blood to flow through them more easily. This may be important in people who have Fontan circulation that lack a ventricle to pump blood through the pulmonary arteries."
The Fontan procedure is part of a series of reconstructive heart surgeries to help reroute blood flow for children with congenital heart disorders affecting one lower chamber of the heart – or single ventricle defects. These patients could have a smaller or underdeveloped chamber or be missing a valve due to heart problems like hypoplastic left heart syndrome or tricuspid atresia.
The trial included 88 enrolled adolescents from more than 20 centers who had undergone a Fontan procedure. Researchers wanted to see if relaxing the blood vessels in the lungs with udenafil would also allow the blood vessels in the liver to relax.
After 12 months, there was no evidence of reduced liver stiffness, or vessels being relaxed or less congested. However, blood tests detected significant decreases in ongoing scarring, or fibrosis, that can lead to more serious liver disease.
Schumacher said the finding warrants a larger, longitudinal study.
"We don't understand yet how or whether this treatment will meaningfully help the liver, but perhaps by relaxing the pulmonary arteries, we are changing something about how congestion affects the liver and leads to scarring," Schumacher said.
"For a disease without treatments, anything that may affect the onset or progression of liver scarring is worth learning more about."
Funding: The trial was funded by the National Heart, Lung, and Blood Institute of the National Institutes of Health with financial support from the sponsor, Mezzion Pharma Co. Ltd.

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!