How researchers, doctors and recovered patients are joining efforts to see if one of the oldest pandemic treatments really works.
4:37 PM
Author |
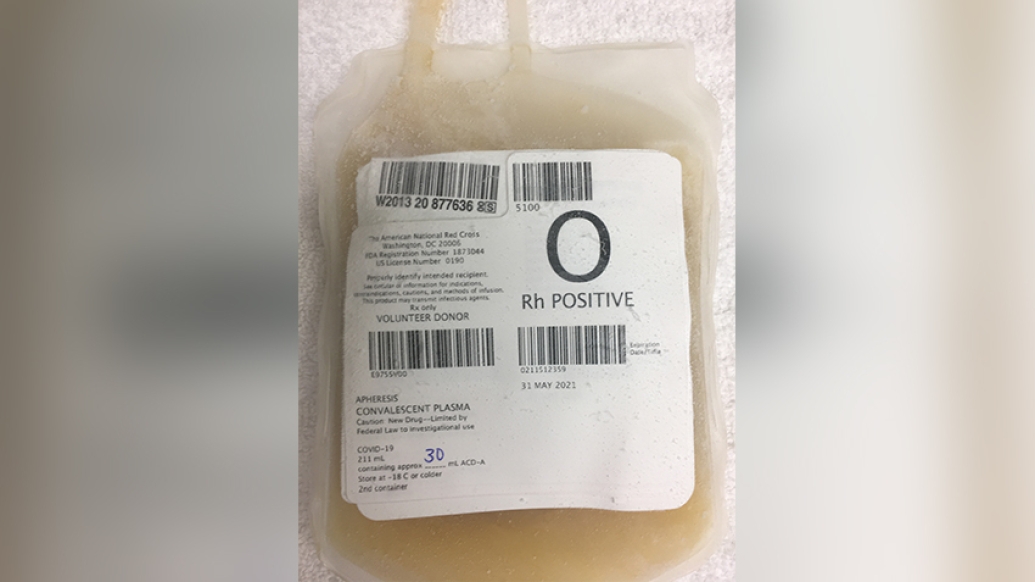
The bag of frozen yellow liquid doesn't look like much, but it might have the power to help a patient survive COVID-19.
Or it might be another promising idea that doesn't quite pan out.
The bag holds convalescent plasma, a golden elixir distilled from the blood of someone who has already recovered from a case of COVID-19.
That person donated something from their own body, hoping to help another. That same hope has inspired countless other donors, in epidemics going back more than 110 years. And the doctors who have infused that plasma into their patients have hoped that transferring its disease-fighting antibodies from a recovered person to a sick person will help.
But hope isn't enough when it comes to fighting a new and deadly virus on a massive scale. That's why researchers and physicians around the world have launched studies of convalescent plasma, to help them get firm, scientific answers about how powerful it really is against COVID-19.
Strictly controlled research could take convalescent plasma from something that has seemed to help people fight off a dangerous infection, to something that we know will help either certain patients, many patients – or no one.
A gold-standard trial for convalescent plasma
One of those studies has a very serious aim, but a light hearted name: C3PO, short for Clinical Trial of COVID-19 Convalescent Plasma of Outpatients. It focuses on people who have early signs of COVID-19 and a high risk of getting seriously ill as time goes on.
Several hospitals, all in Michigan, have started enrolling patients in the C3PO study as they visit the emergency department for their symptoms. Over 50 hospitals nationwide will soon do the same and will enroll 600 patients.
But instead of giving convalescent plasma to every patient who consents to take part, they will randomly assign half to get real plasma, and half to get a saline infusion. Introducing this element of chance, and the saline placebo, makes C3PO a randomized controlled trial – the gold standard for showing cause and effect in medicine.
The study will be conducted in the research organization called Strategies to Innovate Emergency Care Clinical Trials Network, or SIREN, funded by the National Institutes of Health. Researchers at Michigan Medicine, the University of Michigan's academic medical center, run the Clinical Coordinating Center for SIREN.
"Before now, the majority of trials of convalescent plasma for COVID-19 have been in critically ill or hospitalized patients," says Frederick Korley, M.D., Ph.D., an emergency medicine physician and one of the principal investigators for C3PO. "But antiviral therapies may be most effective when given early during the course of illness, prior to significant organ damage. We're trying to do that by identifying people who are mildly ill, but at high risk, with the hopes that we can neutralize the virus before it can cause irreparable harm.
"The question our study seeks to ask is whether we can prevent patients from getting sicker, requiring emergency care, urgent care or hospitalization, within 15 days of receiving convalescent plasma," he explains.
Korley and his colleagues at U-M, the University of Pittsburgh, the Medical University of South Carolina and Stanford University set a minimum concentration, or titer, of antibodies that the plasma must contain to be used in C3PO.
Different donors may have different titers of antibodies naturally, based on how their bodies reacted to the virus and how long it's been since they were sick. The study will track the titers of antibodies each patient receives, and see if there's any difference in how well they fare.
Korley's colleague William Barsan, M.D., an emergency physician at Michigan Medicine who helps oversee SIREN, calls the study pivotal. "There's never been a randomized controlled trial that shows that convalescent plasma can prevent worsening of patients with any infectious disease," he says. "There have been suggestions, but really no proof."
Tracking thousands of inpatients in the C3PO clinical trial
C3PO, and a few other randomized clinical trials that have already started or are getting ready to launch, will provide a high level of scientific evidence to evaluate the effectiveness of convalescent plasma in COVID-19 infection.
Ongoing at the same time is a massive effort to collect and study data from hospitalized COVID-19 patients who have received convalescent plasma in the U.S. since the pandemic began.
Michigan Medicine is one of more than 2,700 hospitals that are taking part in the US COVID Plasma effort.
Before now, the majority of trials of convalescent plasma for COVID-19 have been in critically ill or hospitalized patients. But antiviral therapies may be most effective when given early during the course of illness, prior to significant organ damage.Frederick Korley, M.D., Ph.D.
So far, nearly 87,000 patients nationwide have received plasma through the program, which is approved by the FDA as an "expanded access" program – a way for patients to receive an experimental therapy outside of a clinical trial. Like C3PO, it's funded by the Biomedical Advanced Research and Development Authority, or BARDA, which focuses on potential treatments for emerging diseases.
While the effort won't produce as rigorous of results as a trial would because it doesn't randomly assign patients to get plasma or not, it could still yield good evidence about the safety and side effects of convalescent plasma treatment.
SEE ALSO: Hospitals Can't Get Back to Normal Without More Blood. Here Are 7 Ways You Can Help
Robertson Davenport, M.D., the pathologist who directs Michigan Medicine's Blood Bank, has overseen the use of convalescent plasma at his health system. He also emphasizes using it early in a patient's hospitalization, before their organs are too damaged by the body's overreaction to the virus.
Patients and their families should ask about it early on, especially if they're being transferred to another hospital. "Many of the patients who were transferred to our hospital during Michigan's peak arrived too late for convalescent plasma to have an effect, because their organs were already too damaged," Davenport says.
He worries about the potential effects if the U.S. Food and Drug Administration grants permission for doctors to prescribe convalescent plasma outside of the expanded access and clinical trial pathways.
While this might increase access, it could also dilute the scientific effort to measure convalescent plasma's true power because individual doctors won't have to report data for analysis. And hospitals will have to pass along costs for each unit of plasma to insurers or patients, which they currently don't need to do.
However, Korley notes, if a patient is offered plasma through a trial such as C3PO, or a hospital taking part in the large registry, that's still going to be their best chance of getting it for the near future. And even if someone enters the C3PO trial and gets the placebo in the emergency room, they can still be eligible to get convalescent plasma if they have to be hospitalized.
Urgent need for COVID-19 convalescent plasma donors
People who are giving convalescent plasma should do so as early as possible after their symptoms are gone, Davenport says. That way their antibody levels are as high as they might get, but their blood won't contain the circulating virus.
Right now, someone must be fully recovered from their COVID-19 symptoms and have had a confirmed test for the virus to give plasma. So people who were sick during the peak months in March through June in Michigan should give as soon as possible. Donors can give every 28 days.
"This plasma lasts a year in the freezer, and so by donating now people can help us get ahead of what might be coming this fall and winter," Davenport says.
People who once had to wait a year before giving any sort of blood product – such as people who had traveled to certain countries, gotten a tattoo or piercing, or had certain kinds of sexual contact – now only have to wait three months.
Someone who never had a test for COVID-19 can still see if they have antibodies against it, by donating whole blood or platelets through the Red Cross and getting a free test. That could also help the effort to understand how antibody levels change over time after someone recovers. But at this time, someone identified as having antibodies this way won't be asked to give convalescent plasma.
Getting all the answers we can about convalescent plasma during the COVID-19 pandemic, in the most scientific way possible, could help us be ready for the next pandemic, Davenport notes.
"The idea of passive immunity through plasma transfusion has been around for over a century, and there's good historical data for it. But it's not at the level of evidence we can get now," he says. "And we know that there will be more viruses after this one. Let's prove it one way or another for COVID-19 with C3PO and other clinical trials, so we're ready for the next one.

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!