Under development at C.S. Mott Children’s Hospital, the pediatric artificial lung could offer new hope for treating children with lung failure.
1:00 PM
Author |
On any given day in the pediatric and neonatal intensive care units at University of Michigan C.S. Mott Children's Hospital, there might be one or two children hooked up to pumps and artificial lungs keeping them alive.
LISTEN UP: Add the new Michigan Medicine News Break to your Alexa-enabled device, or subscribe to our daily audio updates on iTunes, Google Play and Stitcher.
Connected to the machines, these young patients are usually bedridden. Beeps are the soundtrack to their days; a web of wires forms one of their earliest views.
That all could change with implantable, simpler artificial lung systems that can support patients while they play outside or eat dinner at their families' dining room tables.
Ronald Hirschl, M.D., and his team at Michigan Medicine are developing the pediatric artificial lung (PAL) — technology that is designed to allow patients experiencing lung failure to go anywhere with artificial lungs on their backs.
"As soon as five years from now," says Hirschl, the head of pediatric surgery at Mott, "treatment for these patients will look vastly different from what it is today."
His research team recently received a $2.6 million grant from the National Institutes of Health to develop the PAL and test it for clinical use. It is designed to be implanted and to reside outside of the body, attached to the large blood vessels near the heart and to the heart itself. Research and testing will continue for the next several years.
"Developing this lung would be a significant advance in what is now a very complex process for caring for these patients," says Hirschl. "You simplify it so much."
The PAL's evolution is similar to that of the artificial heart first implanted in 1982. It was hard to imagine 20 years before that time, Hirschl says, that an artificial heart — one that could work indefinitely — was possible. Now, it's a common treatment.
"We are moving towards developing this so we can do the same thing," Hirschl says.
Lifesaving potential, portability
The portable lung could dramatically change the lives of many of the thousands of patients who require support from a device in a treatment called extracorporeal membrane oxygenation (ECMO).
Currently, when pediatric and adult patients experience heart or lung failure, a pump and artificial lung provide support using ECMO, which was developed in the 1970s by U-M surgeon Robert Bartlett, M.D.
MORE FROM THE LAB: Subscribe to our weekly newsletter
The number of patients hooked up to ECMO has skyrocketed over the past decade — increasing more than fourfold between 2005 and 2016, according to the Extracorporeal Life Support Organization. Among the reasons for this is the increase in the number of patients who have contracted H1N1 pneumonia.
ECMO uses a pump and an artificial lung to take over the function of the heart or lungs long enough for those organs to recover or ultimately be replaced with a transplant.
But patients hooked up to ECMO must stay in the intensive care unit, where they lie in bed, often immobile — sometimes for weeks or months. This, Hirschl says, can lead to muscle atrophy and a decline in physical condition that can reduce the ability to survive with or without a lung transplant.
Years of clinical experience have shown that when patients have been allowed to exercise, they are in better physical condition when they undergo transplantation of other organs, such as a heart.
That's where the PAL could be lifesaving.
Developing this lung would be a significant advance in what is now a very complex process for caring for these patients.Ronald Hirschl, M.D.
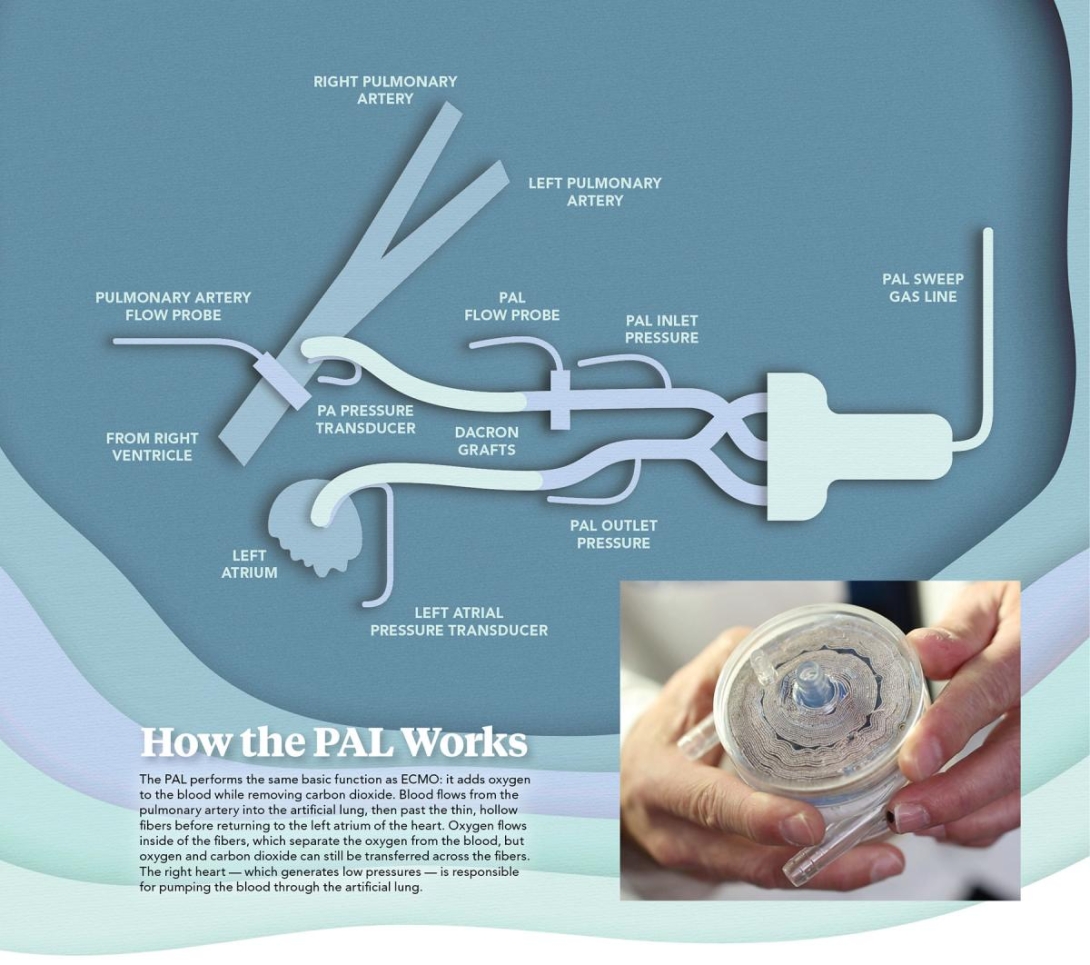
How the PAL works
The PAL, which is about the size of an adult's hand and is made of hollow fiber membranes, performs the same basic function as ECMO: it adds oxygen to the blood while removing carbon dioxide.
SEE ALSO: Artificial Placenta Holds Promise for Extremely Premature Infants
Blood flows from the pulmonary artery into the artificial lung, then past thin, hollow fibers before the blood returns to the left atrium of the heart. Oxygen flows inside of the fibers, which separate the oxygen from the blood. But oxygen and carbon dioxide are transferred across the fibers.
The right side of the heart — which generates low pressures — is responsible for pumping the blood through the artificial lung. The artificial lung accommodates these pressures by using low resistance.
Michigan Medicine, which has successfully demonstrated the PAL's efficacy in the laboratory, is one of just a few medical centers developing this type of artificial lung, Hirschl says. His group has been funded to study an implantable artificial lung for the past 15 years.
Hirschl says a big advantage with the PAL system is that blood thinners, which currently are required with ECMO and can lead to bleeding complications, may not be required. The team is experimenting with coating the circuits with different types of anticoagulants that would reduce blood clotting.
One of the PAL's most promising features is that it allows time for the lung to heal, which may eliminate the need for a lung transplant altogether.
Bartlett, an emeritus professor of surgery who has been participating in the project — and has been involved in the field of ECMO since 1965 — explains that research shows that prolonged support from an external system can allow lungs to heal and recover back to normal.
That is far better than having to endure a transplant, which requires waiting for a donor and then taking immunosuppressant drugs for life and enduring chronic lung problems such as pulmonary fibrosis, he says.
But he explains that regeneration could take months, so it's not feasible with the current ECMO system, which forces the patient to remain in the ICU, a complicated and expensive endeavor. ECMO is usually turned off after 30 days if the patient doesn't show improvement.
"We are aiming to simplify our approach so that we can manage patients out of the ICU and even at home," Bartlett says.
The road ahead
Bartlett and Hirschl agree that the day when a patient could go home with a PAL is roughly five years away, but challenges remain in developing the technology.
For example, when patients are active and exercising, they produce more carbon dioxide. A mechanism needs to be developed to increase the amount of carbon dioxide removed by the artificial lung during those times.
SEE ALSO: Lab-Grown Human 'Mini' Lungs Successfully Engraft in Mice
In addition, monitoring and alarm systems are necessary to notify the patient and doctors when the artificial lung is failing and needs to be changed.
During the early stages of development, it is likely that patients with the lung backpack will begin in the ICU and then walk around the hospital floor, Hirschl says. But the goal is that patients eventually will be able to go home.
Implanting it internally could be years or decades down the road, he says.
"The PAL is an extension of the work of so many physicians, such as Dr. Bartlett, who have contributed to developing lifesaving techniques like ECMO," Hirschl says. "The work that is being done with the PAL will allow such lifesaving approaches to be applied more broadly and effectively and revolutionize how we approach patients with lung failure."
This article originally appeared in Medicine at Michigan magazine.

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!