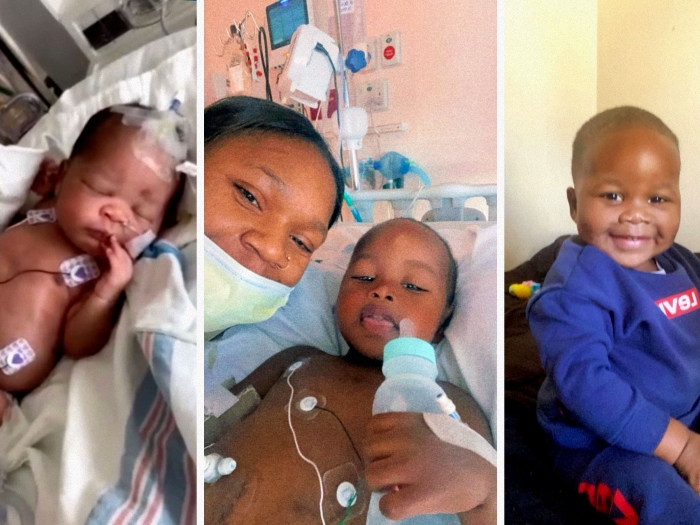
The development of an artificial placenta — used successfully in premature lambs — could revolutionize the treatment of extreme prematurity.
5:04 PM
Author |
Researchers at the University of Michigan are working to improve survival rates in the tiniest, most premature babies in a groundbreaking way: through an artificial placenta that mimics the womb.
MORE FROM THE LAB: Subscribe to our weekly newsletter
The technology hasn't reached a clinical trial, but researchers from U-M's C.S. Mott Children's Hospital and Extracorporeal Circulation Research Laboratory are making dramatic progress. An extracorporeal artificial placenta at the institution has kept five extremely premature lambs alive for a week. The lambs were transferred to the artificial placenta, which utilizes extracorporeal membrane oxygenation (ECMO), without ever taking their first breath.
The ultimate goal of nearly a decade of sustained work: for an artificial placenta to help extremely premature babies with the greatest risks of disability or death continue critical organ development outside of their mother's womb.
Our research is rapidly progressing. Given our recent advances, we believe that the artificial placenta may be used in premature babies in the next five years.George Mychaliska, M.D.
'A complete paradigm shift'
Despite significant advances in the treatment of prematurity, the risk of death and long-term disability remains high for extremely premature infants (born before 24 weeks). Their bodies simply are not prepared for life outside the womb.
SEE ALSO: Building a Better Feeding Tube
"One of the gravest risks for extremely premature babies is undeveloped lungs that are too fragile to handle even the gentlest ventilation techniques," says George Mychaliska, M.D., the principal investigator and the director of U-M's Fetal Diagnosis and Treatment Center. "If a baby's lungs are severely immature, they cannot provide the brain, heart and other organs the oxygen they need to survive."
Mychaliska, who has been referred to as Michigan's "fetus fixer" for his renowned fetal intervention work, has been leading research to improve outcomes for premature infants.
"We thought, 'Why don't we solve the problem of prematurity by re-creating the intrauterine environment?'" he says. "Maybe we should treat this tiny baby like a fetus. Maybe we should treat these babies as if they are still in the womb. This is a complete paradigm shift. Our research is still in a very preliminary stage, but we've passed a significant milestone that gives us promise of revolutionizing the treatment of prematurity.
"Although many of our current therapies are lifesaving, they are not designed for premature babies and are often ineffective or contribute to complications," he adds.
How it works
The innovative artificial placenta simulates the intrauterine environment and provides gas exchange without mechanical ventilation. By recapitulating normal fetal physiology to re-create the intrauterine environment, the artificial placenta holds the promise of normal growth and development outside the womb for extremely premature infants until they are ready for postnatal life.
The success of keeping lambs alive through this technique was a crucial milestone in securing a $2.7 million R01 National Institutes of Health grant to accelerate this research.
The way forward
Over the next five years, researchers expect to demonstrate that an artificial placenta can simulate the intrauterine environment and support a fetal lamb from extreme prematurity to normal newborn physiology. The next step would be to determine if the milestones would justify preliminary clinical trials in extremely premature babies.
SEE ALSO: Developmental Differences in Late Preemies May Not Emerge Until After Age 2
It's a vision first imagined nearly 50 years ago shortly after the advent of cardiopulmonary bypass, when researchers described the potential of a future artificial placenta in a Life Magazine story.
U-M's Robert Bartlett, M.D., known as the "father of ECMO" for developing the lifesaving heart-lung technology, is also a contributor to the lab's work.
"With a better understanding of the physiology of prematurity and advanced technology, we are now in a position to bring this innovative approach envisioned five decades ago to fruition," says Bartlett, a professor emeritus of pediatric surgery.

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!