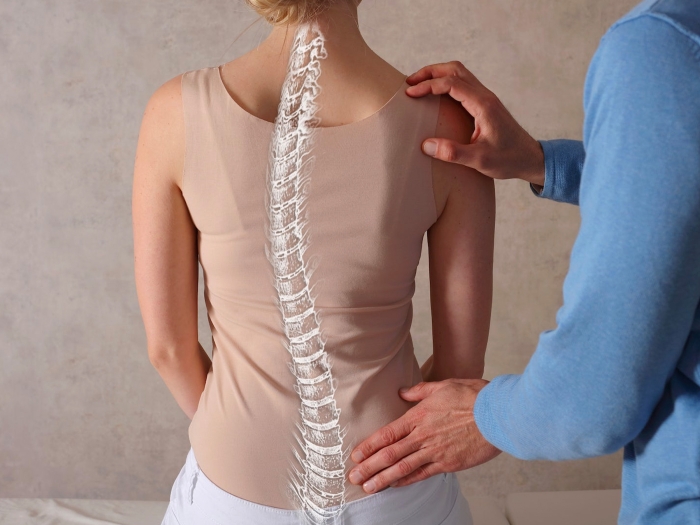
Researchers identified two novel genes that affect bone-formation cells relevant to fractures and osteoporosis; understanding these genes could lead to more effective treatments.
2:36 PM
Author |
Examining a person's DNA can help scientists understand if the individual is at risk for osteoporosis, the chronic bone-weakening condition that affects millions of Americans.
In a new study, published in Nature Communications, researchers used data analysis tools and three-dimensional studies of genomic geography to identify new risk genes for osteoporosis.
"Identifying a disease's actual underlying cause often helps to steer us toward correct, targeted treatments," says Struan F.A. Grant, Ph.D., co-leader of the study and a director of the Center for Spatial and Functional Genomics at Children's Hospital of Philadelphia.
"We have identified two novel genes that affect bone-forming cells relevant to fractures and osteoporosis. Furthermore, the research methods we used could be applied more broadly to other diseases with a genetic component."
Grant's co-leaders on the study include Kurt D. Hankenson, D.V.M., Ph.D., professor of orthopaedic surgery and a researcher in bone formation and remodeling at Michigan Medicine, and Andrew D. Wells, Ph.D., an immunology researcher at CHOP and fellow director of the CSFG.
"Understanding these causative genes could lead to more effective treatments for osteoporosis in the future," Hankenson says.
Examining genetic loci
The study team investigated genetic loci, or DNA regions, previously established in genome-wide association studies (GWAS) to be associated with bone mineral density, both in adults and children.
"Scientists have known for some time now that the nearest gene to a variant associated with a disease is not necessarily the cause of the disease," Wells says.
This suggests to us that follow-up studies investigating biological pathways affected by this gene may present targets for therapies to strengthen bone mineral density and ultimately prevent fractures.Kurt Hankenson, D.V.M., Ph.D.
Because GWAS research detects single-base changes in DNA that are usually not located in obvious parts of the genome, much research has turned to the broader context of interactions within the genome —the entire complement of DNA within cells.
Sometimes the changes, called single-nucleotide polymorphisms, or SNPs, found in GWAS are located close to a culprit gene. More often, the signal comes from a non-coding region of the DNA that regulates another gene that may be thousands of bases away on the DNA sequence.
"The geography of the genome is not linear," Grant says. "Because DNA is folded into chromosomes, parts of the genome may come into physical contact, enabling key biological interactions that affect how a gene is expressed. That's why we study the three-dimensional structure of the genome."
In analyzing how chromatin, the fibers that make up chromosomes, is arranged into specific shapes, spatial genomics offer insight into how genes physically interact with the regulatory regions in DNA that initiate transcription. Transcription is the process in which DNA is copied into RNA, the first event in gene expression.
The research team used high-resolution tools to analyze genome-wide interactions in human osteoblasts—bone-forming cells derived from mesenchymal stem cells. Their analytical tools use a "multi-omic" approach, integrating data from the genome sequence and details of chromatin structure to map interactions between potential BMD-related gene promoters and regions harboring genetic variants related to BMD biology.
Pinpointing genes
The study pinpointed two novel genes, ING3 and EPDR1, which in turn revealed strong effects on human osteoblasts.
"While we're not ruling out other possible causative genes in these regions, the ING3 gene particularly stood out because we found that the genetic signal at this region was the strongest one associated with bone density at the wrist—the major site of fracture in children," says Alessandra Chesi, Ph.D., first author of the study and a researcher at CHOP.
Hankenson notes, "This suggests to us that follow-up studies investigating biological pathways affected by this gene may present targets for therapies to strengthen bone mineral density and ultimately prevent fractures."
On-going studies by Yadav Wagley, Ph.D., a researcher in Hankenson's laboratory, are investigating the pathways affected by INGR3 and EPDR1.
Researchers have long known that bone accrual during childhood may bolster bone health in adulthood. The research team hopes this line of research may present strategies to build upon that knowledge.
Grant adds that the analytical approach used in this study could have a broader use in studying other genetic diseases – including pediatric conditions. His team is collaborating with researchers at CHOP and other centers to generate atlases of promoter-variant interactions across multiple cell types.
"Our hope is that this approach and these atlases could offer up great resources in crafting targeted treatments for many different diseases, including certain pediatric cancers, diabetes and lupus," he says.
The National Institutes of Health (grant HG010067) supported this research.
John Ascenzi of CHOP contributed to this report.

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!