Research reminds physicians that drugs their patients commonly use — from antibiotics to herbal supplements — may cause liver injury or failure.
8:00 AM
Author |

Patients' good intentions to get over a respiratory infection or an ear infection or to lose weight can go wildly wrong if the drug they're taking damages the liver.
New studies remind physicians that drugs their patients commonly use, such as antibiotics and herbal and dietary supplements, are infrequent causes of liver injury and failure. Warnings about liver damage caused by taking too much acetaminophen (Tylenol) have been on over-the-counter packages for years, but other prescription drugs can also lead to severe liver injury no matter the dose or how long the person has taken it.
"Most people with drug-induced liver injury recover fully, but one-quarter of patients will die, need a liver transplant or have lingering liver problems six months after onset," says Robert Fontana, M.D., liver specialist in the University of Michigan Hepatology Program.
Fontana is an investigator in the national Drug-Induced Liver Injury Network (DILIN) that's tracking troubling trends and looking for biomarkers that may make some of us vulnerable to drug-induced liver damage.
While the medicine may be effective, genetic markers may suggest that for some patients another medication or therapy is better and safer to take.Robert Fontana, M.D.
Trends in supplements
The Drug-Induced Liver Injury Network is studying hepatotoxicity caused by both conventional medicines prescribed by doctors and over-the-counter herbal and dietary supplements. Liver injury caused by herbal and dietary supplements increased from 7 percent to 20 percent from 2004, when the group's work began, to a decade later.
The significant increase in liver injury cases attributed to herbal supplements is a phenomenon that's largely unchecked by regulators and doctors. As Fontana explains, most doctors don't know if patients are taking supplements unless patients tell them. Herbal supplements are not routinely reviewed by regulators.
Data shows that half of U.S. adults take an herbal supplement and that the use of complementary and alternative medicines has been increasing in Western countries.
Herbal drugs and supplements including kava, bodybuilding supplements, green tea extract and some weight loss supplements containing usnic acid have a well-known potential to cause liver injury, according to the DILIN.
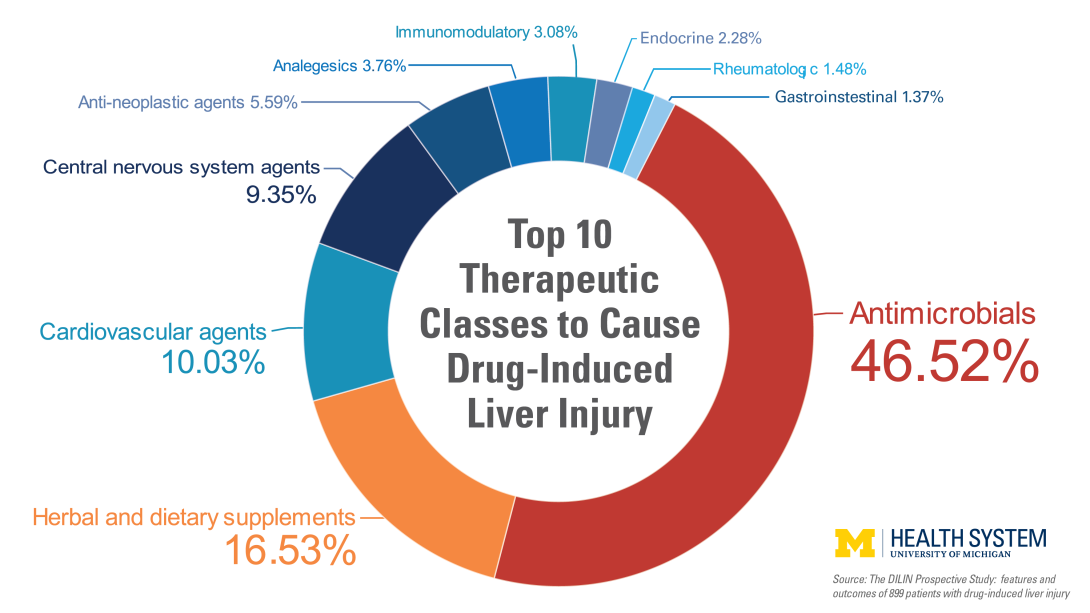
Features and outcomes of 899 patients with drug-induced liver injury: DILIN Prospective Study.
Who's at risk?
Although many classes of drugs can damage the liver, antibiotics, used to treat a variety of bacterial infections on a daily basis, are the most common class and account for 45 percent of cases. Liver injury caused by drugs such as azithromycin (known as Z-Pak) and amoxicillin/clavulanate (Augmentin) can occur within one to three weeks.
Patients with drug-induced liver injury may experience nausea, itchy or yellowing skin, and abdominal pain and have dark urine. The frequency of harm is low compared with the very large number of prescriptions written each year, but population-based estimates suggest injury or damage occurs at a frequency of 1 to 10 per 100,000 drug prescriptions.
"This form of liver injury is frequently not seen during the drug development process due to its low incidence and therefore may not be included in a product insert for several years until multiple bona fide cases are reported," says Fontana, who's collaborating with infectious disease specialists and pediatricians to raise awareness of unexplained liver injury. "Therefore, many patients and providers may be unaware of the potential risk of liver damage from commonly prescribed drugs."
Investigators in DILIN are looking at age, gender and the reason patients were taking the drug in the first place to determine who's at risk for drug-induced liver injury. The DILIN is also creating a bank of biological specimens consisting of DNA, plasma and immortalized lymphocytes to look for a common genetic risk factor among the more than 1,000 study participants.
"It can lead to a second tier of decision-making," says Fontana. "While the medicine may be effective, we hope to develop 'personalized' genetic markers that may suggest that for some patients another medication or therapy is better and safer to take."

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!